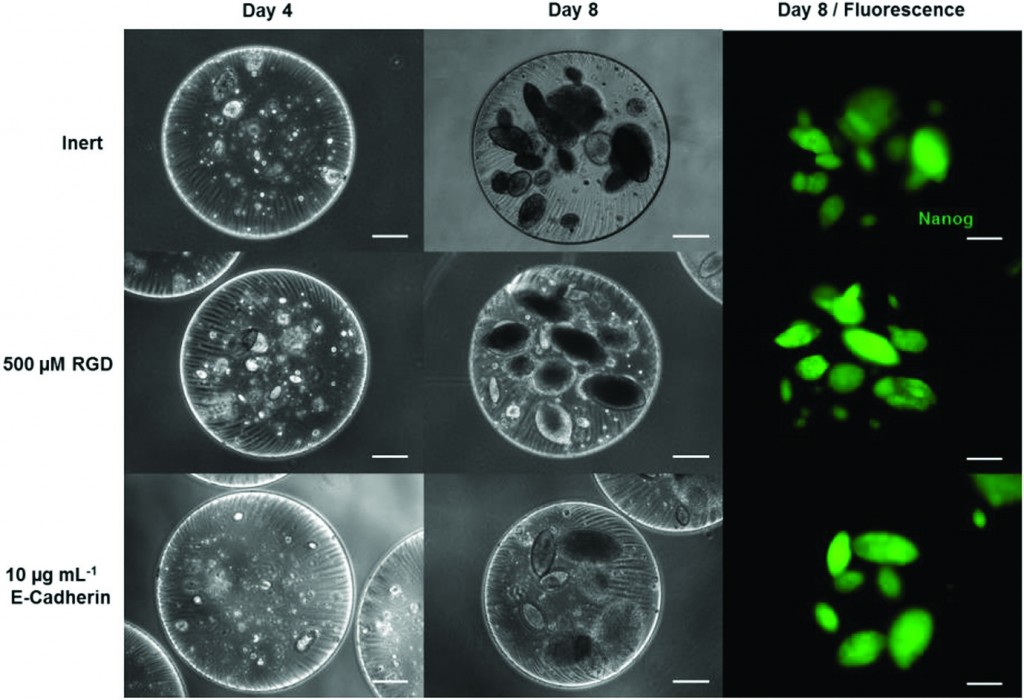
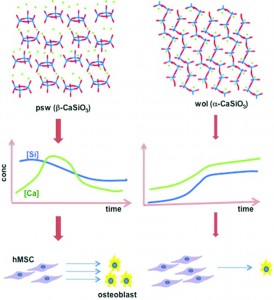
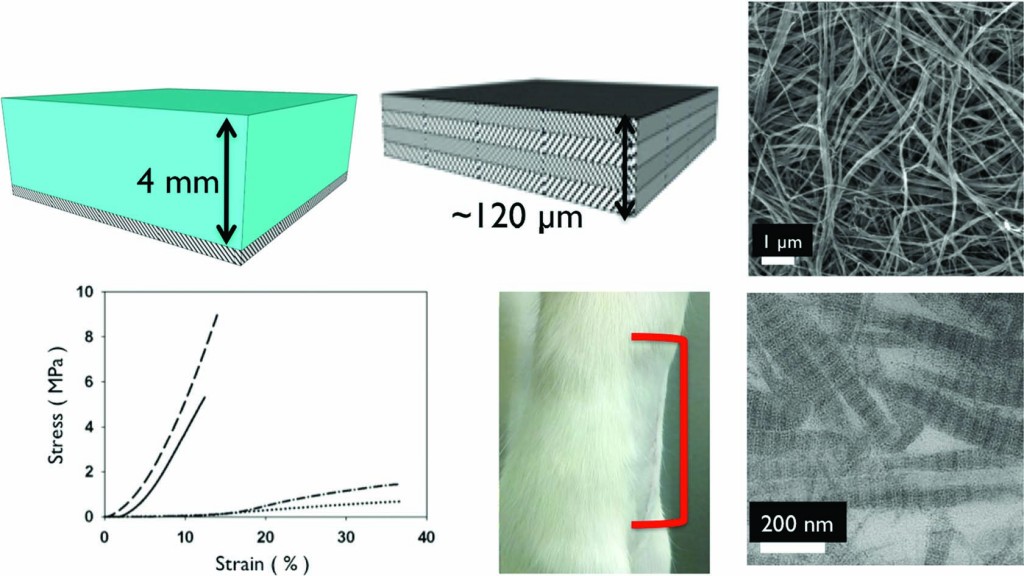
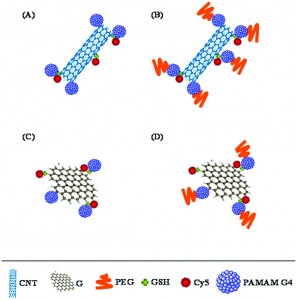
The next generation of diagnostic devices is currently being realized in the form of ‘smart’ biosensors. These sensors use logic gates to digitalize biochemical signals and produce a binary output that either indicates the presence or absence of a disease. The biochemical signals, which are often the product of enzymatic cascades, are also capable of triggering drug release from stimuli-responsive materials, thus creating a device that is capable of both sensing and treating a particular condition.
However, these integrated sense and treat biosensors have a major obstacle to overcome if they are to be successfully implemented. Many of the enzymatic cascades used for input will produce low, but non-zero concentrations of the sensing analytes under normal, physiological conditions. Over time, the accumulation of these background signals can rise to a level that is high enough to trigger drug release, resulting in the dosage of a perfectly healthy patient which often has adverse consequences. In this work, a team from Clarkson University and Oak Ridge National Laboratory propose a novel solution to this problem which involves the incorporation of an enzymatic filter into these devices.
The group created a system that was designed to detect and treat liver injury using input from two known biomarkers of liver disease: alanine transaminase (ALT) and aspartate transaminase, type I (AST). When both ALT and AST are present in elevated concentrations, they will produce high levels of molecules which can be used to synthesize citric acid and another enzyme known as CoA-SH. While CoA-SH can be readily detected and used to generate a positive signal indicating liver injury, citric acid can be used to trigger drug release by dissolving the iron-cross-linked alginate beads which were used in this study to simulate drug release capsules. The group then introduced an enzymatic filter, malate dehydrogenase (MDH), into the system. MDH is able to regulate the production of citric acid, thus controlling the degradation of the alginate beads and subsequent drug release. In the test cases listed, the modified system generated the correct diagnostic result 100% of the time. However, the filter was not able to completely eliminate bead dissolution and drug release under conditions associated with a normal, healthy environment.
To tackle this problem, the group decided to try improving the stability of the beads themselves. By alternately depositing layers of poly-L-lysine with additional layers of alginate, PLA bi-layers were formed on the surface of the alginate cores. The group found that, as more layers were added, the speed of bead dissolution and the amount of drug released were reduced. The combination of the filter and the PLA coatings showed enhanced suppression of the negative signals while continuing to have a minimal effect on the positive signal.
Although the group was unable to completely eliminate drug release under normal conditions due to long-term noise accumulation, they were able to reduce it significantly. Their approach marks a promising step forwards in addressing one of the major obstacles faced by smart biosensor systems.
Enzymatic filter for improved separation of output signals in enzyme logic systems towards ‘sense and treat’ medicine
Shay Mailloux, Oleksandr Zavalov, Nataliia Guz, Evgeny Katz and Vera Bocharova
Biomater. Sci., 2014, Advance Article DOI: 10.1039/C3BM60197H
Ellen Tworkoski is a guest web-writer for Biomaterials Science. She is currently a second year Ph.D. student in the biomedical engineering department at Northwestern University (Evanston, IL, USA).
Follow the latest journal news on Twitter @BioMaterSci or go to our Facebook page.
To keep up-to-date with all the latest research, sign-up to our RSS feed or Table of contents alert.