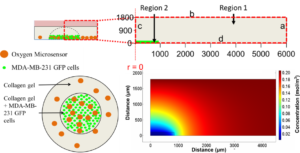
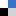Microenvironmental oxygen levels and gradients within three-dimensional (3D) tissue cultures directly influence cellular behavior and function, dictating the mode of proliferation, metabolism and interaction of cells with each other and their environment. While advances and prevalence of in vitro generated 3D cultures have spurred new techniques and systems for biological interrogation, it is necessary to develop and implement parallel systems to monitor and characterize the oxygen microenvironment within the tissue cultures and around them in the vessel used for the cultures. Conventional oxygen evaluation platforms can be ill-suited for continuous oxygen evaluation in custom tissue cultures. The Takayama group was able to robustly evaluate multiple 3D culture platforms by combining the use of phase-fluorimetry and lab-fabricated dispersible oxygen responsive microparticles. Oxygen microsensors were used to evaluate two spheroid culture vessels, hanging-drop and low-adhesion microwell plates, to highlight the variations in the oxygen levels peripheral to the spheroids in the two culture techniques. Dramatic differences can be seen in the steady state oxygen levels between the two culture techniques because of the difference in distance between the spheroids and the air-liquid interface in these two vessel types. These results highlighted the importance of minding the gas exchange location as compared to the cell culture to ensure appropriate tissue culture microenvironments.
Furthermore, these microsensors were used to map radial oxygen distribution across a circular, cell-patterned hydrogel by dispersing the microsensors within the culture. Coupling the spatial oxygen mapping to computational models of oxygen diffusion, the authors were able to estimate oxygen uptake behavior of the tissue culture. While 3D tissue culture platforms leverage the in vitro tissue architecture to produce more physiologically similar phenomena, integrated design and analysis of these 3D cell cultures from both biomaterial and oxygen supply aspects will be paramount in enabling researchers to effectively recreate some of the complexities present within both healthy and diseased tissues.
Tips from the authors:
- When fabricating oxygen microsensing beads, infusion with Dichloromethane enabled large amount of Ruthenium caging within the PDMS microspheres, while leaving them oxygen sensitive. While other solvents swell PDMS more readily and enabled higher efficiency infusion of ruthenium, these solvents resulted in oxygen unresponsive ruthenium loaded PDMS beads.
- Microsensors cannot be effectively integrated in the multicellular spheroids we tried with HEK293T, HS-5 and MDA-MB-231 cells; as the spheroids contract microsensors are ejected out of the spheroids.
- The only limitation of phase-fluorimetry for the oxygen measurements is sufficient signal output that it can be detected by the photodiode, or other detection system. This was generally not a problem with beads greater than 80 microns assuming the culture systems was less than 1-mm thick. However, we were unable to effectively infuse beads under 80 microns with enough ruthenium to have enough output signals from the microsensors to get robust readings with cultures > 1 mm.
Article Link:
Dispersible oxygen microsensors map oxygen gradients in three-dimensional cell cultures Biomater. Sci., 2017,5, 2106-2113
Dr. Sudip Mukherjee is a Web Writer for Biomaterials Science. He is currently a Postdoctoral Research Associate working alongside Dr. Omid Veiseh at the Department of Bioengineering at the Rice University. His research is involved in the development of advanced nanomaterials for drug/gene delivery in cancer theranostics, immunomodulatory applications & angiogenesis. He published a total of ~30 research articles/patents. He serves as International Advisory Board Member for ‘Materials Research Express’, IOP Sciences. He is an associate member (AMRSC) of RSC, UK. He serves as reviewer for several international journals like Chem Comm, J Mater Chem A, J Mater Chem B, Journal of Biomedical Nanotechnology, RSC Advances, IOP Nanotechnology etc.