The effective use of earth abundant electrocatalyst copper in splitting of water as nanostructures with different combinations is central in replacing the noble metals for the industrialization of hydrogen generation. Being the carbonaceous fuels as front line suppliers of energy, they adversely lead to affect the environment with the greenhouse gases emission. Considering the electrocatalytic way of splitting water, it is one of the finest ways of producing pure hydrogen with fast rate and assisted with no other undesired by-products and hence researchers across the world focus maximum attention to make it commercially applicable.
To replace the noble metals, transition metals based catalysts are promising and the importance of Cu based nanostructures as effective electrocatalysts for both oxygen evolution reaction (OER) and hydrogen evolution reaction (HER) needs greater attention. Moreover, various synthetic approaches with Cu nanostructures like mono, bi and tri metallic catalysts as oxides hydroxides, sulfides, selenides, tellurides and phosphides were studied for OER and HER in different pH conditions will give a vast analysis on Cu based catalysts towards real scale water splitting electrocatalytically.
Hence, to precise, a brief understanding on Cu-based nanostructures in electrocatalytic water splitting is highly needed to be applied with other advancements in catalysts development for the viable hydrogen generation with electrocatalytic water splitting.
Recently, Karthick. K and Subrata Kundu and co-authors have highlighted the nanostructures of Cu based oxides, hydroxides, sulfides, selenides, tellurides and phosphides as catalysts for water splitting application and the merits of which have been explored in detail as a review. The possibilities of enhancing the activity and durability with Cu based catalysts were studied and among which enhancing the activity with the formation of nanostructures, growing over 3D conducting supports, enhancing the electrical conductivity with graphene, increasing the metallic active sites by different methods like electrodeposition, adding other transition metals and also by varying the stoichiometric ratios had resulted in ensuring astonishing activity and stability.
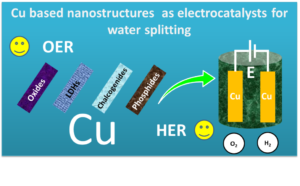 Figure 1. Cu nanostructures as catalysts for enabling high scale OER and HER activities by studying them as oxides, LDHs, chalcogenides and phosphides.
Figure 1. Cu nanostructures as catalysts for enabling high scale OER and HER activities by studying them as oxides, LDHs, chalcogenides and phosphides.
Authors:
CSIR – Central Electrochemical Research Institute
K. Karthick had received his B.Sc degree from Government Arts and Science College, Udumalapet, India and M.Sc degree from The American College – Madurai, India in general chemistry. He qualified UGC-JRF in December-2014 and joined under Dr. Subrata Kundu’s research Group since August-2015. He is currently working towards his Ph.D thesis mainly focused on Electrocatalytic Water splitting applications.
https://scholar.google.co.in/citations?user=RU1m2ScAAAAJ&hl=en
Dr. Subrata Kundu
CSIR – Central Electrochemical Research Institute
Dr. Subrata Kundu received his Ph.D from the Indian Institute of Technology (IIT), Kharagpur, India in early 2005. Then he moved to University of Nebraska, Lincoln, USA and later to Texas A&M University, College station, Texas, USA as a post-doc fellow (from 2005 to 2010). He is currently working as a Senior Scientist at CSIR-CECRI, Karaikudi, India. Dr. Kundu is serving as an editorial board member of several international journals including prestigious ‘Scientific Reports’ from Nature publishers since 2015. Dr. Kundu and his co-workers are working in the forefront area of Material Sciences with emphasizes on energy, environment, catalysis and electrocatalysis.
https://scholar.google.co.in/citations?user=4siGGnQAAAAJ&hl=en
https://www.cecri.res.in/Profile?empcode=40257
https://anantharaj1402.wixsite.com/dr-sk-group
Article information:
Investigation on Nanostructured Cu Based Electrocatalysts for Improvising Water Splitting: A Review
Karthick Kannimuthu, K. Sangeetha, S. Sam Sankar, Arun Karmakar, Ragunath Madhu and Subrata Kundu
Inorg. Chem. Front., 2020, Accepted Manuscript
https://doi.org/10.1039/D0QI01060J