 On the outside cover of Issue 21 we have a hot perspective article from Shinji Yamada and John S. Fossey describing nitrogen cation-π interactions in asymmetric synthesis and summarises the areas where these interactions play an important role.
On the outside cover of Issue 21 we have a hot perspective article from Shinji Yamada and John S. Fossey describing nitrogen cation-π interactions in asymmetric synthesis and summarises the areas where these interactions play an important role.
Areas covered:
- Iminium–π interactions
- Pyridinium–π interactions
- Imidazolium, thiazolium and related structure–π interactions
Nitrogen cation–π interactions in asymmetric organocatalytic synthesis
Shinji Yamada and John S. Fossey
Org. Biomol. Chem., 2011, 9, 7275-7281
DOI: 10.1039/C1OB05228D
 The inside cover highlights work from Radek Cibulka and Tomas Kraus et al. who have used four alloxazine–cyclodextrin conjugates as catalysts for enantioselective oxidation of prochiral sulfides to sulfoxides. Their method avoids the problems of overoxidation to sulfones that sometimes occur when transition metal catalysts are used, and provides better enantioselectivity (up to 77% ee) than other sulfoxidation organocatalysts.
The inside cover highlights work from Radek Cibulka and Tomas Kraus et al. who have used four alloxazine–cyclodextrin conjugates as catalysts for enantioselective oxidation of prochiral sulfides to sulfoxides. Their method avoids the problems of overoxidation to sulfones that sometimes occur when transition metal catalysts are used, and provides better enantioselectivity (up to 77% ee) than other sulfoxidation organocatalysts.
Alloxazine–cyclodextrin conjugates for organocatalytic enantioselective sulfoxidations
Viktor Mojr, Miloš Buděšínský, Radek Cibulka and Tomáš Kraus
Org. Biomol. Chem., 2011, 9, 7318-7326
DOI: 10.1039/C1OB05934C
Both articles are free to access for the next 6 weeks.


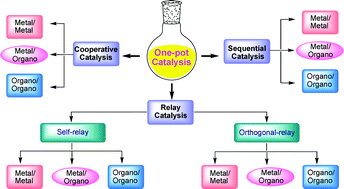









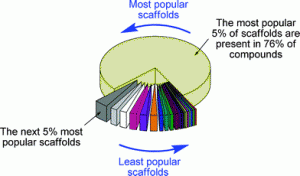
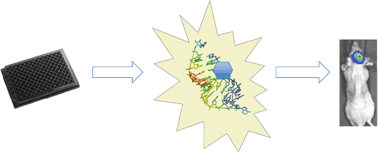
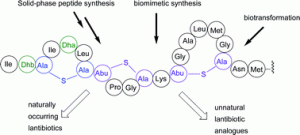



![GA[1]](https://blogs.rsc.org/ob/files/2011/08/GA12.gif)