What inspired you to study science?
During university, I was interested in both the sciences and the humanities, and so I took an advanced English course. After long discussions on different ways to interpret the story, I was frustrated because the instructor would never say whose interpretation was the right one. This frustration solidified that science suited me better, because you can come up with creative proposals and there’s this opportunity to test them and see if they’re valid or not.
What led you to chemistry in particular?
Taking Larry Overman’s sophomore organic chemistry class at the University of California at Irvine was a big turning point for me. Before that I was studying a major between biology and social science, called applied ecology. Larry is an amazing teacher.
A focus of your research is catalysis – could you tell us about a key project you’re running right now in your lab and why you find catalysis so attractive?
One of the major projects in my group right now is focused on catalytic hydroacylation. We want to find ways to selectively activate aldehyde C–H bonds to synthesise ketones, esters and amides. Our goal is to use this C–H activation strategy as a unified approach to all sorts of different heterocycles and polyketides and be able to do this in a way that is regio-, enantio- and chemo-selective. There’s something very attractive about catalysis – you can get things to transform that normally wouldn’t by adding a bit of this magical powder.
What would you say is the major challenge in catalysis?
For my group, the challenge is: how do we bridge that gap from finding something that is novel in reactivity to something that’s going to be wide in applications? It is a difficult challenge, but inspiring to see how catalytic transformations, like metathesis or hydrogenation or cross-coupling, have changed the way people make molecules.
Where do you look for ideas?
I wish there was a journal we could just flip through. Initially I worried that coming up with ideas was impossible, but new ideas pop up all the time through interactions with my students. My students will suggest something and I’ll suggest something else and this going back and forth is what generates and refines our ideas. It’s important for both sides to never shut down the other person’s ideas, but rather build upon them.
One of my students suggested an experiment and instead of saying ‘that’s known with a different catalyst, let’s not do it,’ I said ‘sure, try it and see what happens.’ The result was not what either of us expected. Instead of saying ‘well, this result is interesting but maybe not that interesting,’ we tried to realise the potential in the result, thinking of all the possible ways that we could take it in different directions. That’s how we got started on the ketone hydroacylation project!
Read the full story on Chemistry World.
 Organic synthesis is often heralded as more art than science. An organic chemist’s eye for complexity, breaking down structures into simpler forms, is honed and nurtured over decades. But, is it possible to take this seemingly intangible skill and quantify it, putting a simple number on how complex a chemical structure actually is?
Organic synthesis is often heralded as more art than science. An organic chemist’s eye for complexity, breaking down structures into simpler forms, is honed and nurtured over decades. But, is it possible to take this seemingly intangible skill and quantify it, putting a simple number on how complex a chemical structure actually is?










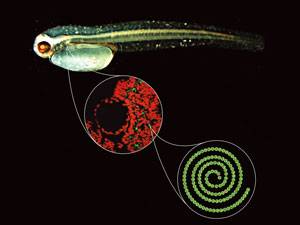
 A new sensing system that
A new sensing system that 
 Oliver Kappe
Oliver Kappe