Want to share your research with like-minded chemists around the world and win prizes without having to leave the lab? Then why not enter our Twitter Poster Conference!
Happening on Monday 20th March (9am GMT) to Tuesday 21st March (9am GMT), this exclusively online event brings members of the scientific research community together to network, share and engage in scientific debate.
How to Take Part
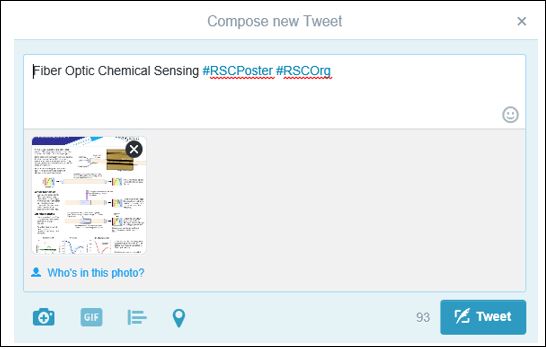
During the event simply tweet an image (e.g. JPEG) which will be a digital poster summarising your research along with #RSCPoster, the most appropriate subject area hashtag (either #RSCOrg or #RSCChemBio) and the title of your work.
For example:
You will then need to upload your poster and details onto our Tumblr site, under either the RSC Org or RSC ChemBio categories depending on your poster (more information on the site).
How to Win
Poster prizes will be given for posters with the best content, design, accessibility and interactions (like questions asked and answered) – so make sure you check back regularly in the day and follow #RSCPoster. There is also an audience award for the most re-tweeted poster.
To be able to win a prize, email us before the event at RSCPoster@gmail.com and let us know your name, contact details, the title of your topic and your twitter ID.
Full terms and conditions can be found here, as well as other subject area hashtags.
GOOD LUCK!