 Synthetic methodologies to control the selectivity and specificity of catalytic reactions remain the subject of intense research due to the importance of chiral compounds in pharmaceuticals, agrochemicals and functional materials. Homogenous catalysis takes advantage of weak interactions between the substrate, catalyst and groups distal to the active site to impart selectivity. Such features are ubiquitous in enzymes which display exceptionally high levels of stereochemical control and activity. In order to exploit the advanced capabilities of such enzymes for selective catalytic transformations, researchers have linked transition metal catalysts with different biological scaffolds–proteins, peptides, DNA–to create artificial metalloenzymes.
Synthetic methodologies to control the selectivity and specificity of catalytic reactions remain the subject of intense research due to the importance of chiral compounds in pharmaceuticals, agrochemicals and functional materials. Homogenous catalysis takes advantage of weak interactions between the substrate, catalyst and groups distal to the active site to impart selectivity. Such features are ubiquitous in enzymes which display exceptionally high levels of stereochemical control and activity. In order to exploit the advanced capabilities of such enzymes for selective catalytic transformations, researchers have linked transition metal catalysts with different biological scaffolds–proteins, peptides, DNA–to create artificial metalloenzymes.
Since their inception in the late 1970’s, artificial metalloenzymes have emerged as a vibrant area of research with numerous examples of a variety of catalytic transformations reported alongside creative methods for incorporating the transition metal complex into the biomolecular scaffold. Key to their design is the ‘second coordination sphere’ provided by the biological scaffold where supramolecular interactions within the active site contribute to the rate and enantioselectivity of the system.
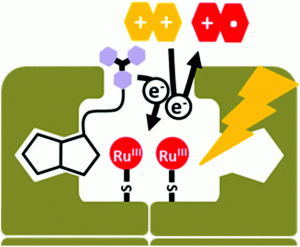
Prof. Thomas Ward and Prof. Oliver Wenger of the University of Basel have recently reported a novel biotin-streptavidin system equipped with an anchored photosensitizer capable of undergoing electron transfers between the biotinylated electron donor and ruthenium(II)-labeled streptavidin.
In the past, studies of the luminescent properties of biotinylated d6 metal complexes, common in photoredox catalysis, have been carried out for the purposes of cell imaging and ruthenium and rhenium complexes have been employed in the elucidation of electron tunneling pathways of various proteins. The application of phototriggered electron transfers to ruthenium photosensitizers, up until this point, had not yet been realized and this recent discovery demonstrates a significant advancement within this field.
This new biotin-streptavidin artificial metalloenzyme contains a ruthenium catalyst and photosensitizers embedded within the biotin binding pocket of streptavidin through covalent interactions with non-native cysteine residues. Their performance in various electron transfer studies demonstrates their potential to behave as advanced photoredox catalysts and given the diversity of reactions amenable to photoredox processes, these novel artificial metalloenzymes will provide unique opportunities within selective catalysis.
To find out more see:
Light-driven electron injection from a biotinylated triarylamine donor to [Ru(diimine)3]2+-labeled streptavidin
Sascha G. Keller, Andrea Pannwitz, Fabian Schwizer, Juliane Klehr, Oliver S. Wenger and Thomas R. Ward
DOI: 10.1039/C6OB01273F
Victoria Corless is currently completing her Ph.D. in organic chemistry with Prof. Andrei Yudin at The University of Toronto. Her research is centred on the synthesis of kinetically amphoteric molecules, which offer a versatile platform for the development of chemoselective transformations with particular emphasis on creating novel biologically active molecules.










