The number of deaths due to heroin overdoses has severely increased in last decade, and regular consumption of heroin leads to serious health concerns.
Vaccination is one of the emerging choices among heroin rehabilitation therapies. In general, these vaccines contain heroin-like hapten as a pharmacophore that acts as an antagonist. This antagonist has high affinity for heroin receptors but does not activate them, hence reverses the effects of heroin. Because of inherent chemical instability, when entered in to bloodstream these anti-heroin drugs rapidly metabolise into several by-products, which makes development of effective vaccines against heroin particularly challenging.
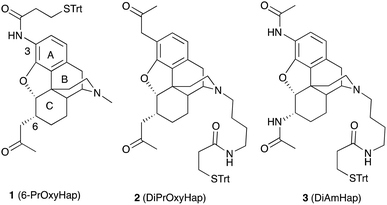
Recently an American research group at National Institute on Drug Abuse (NIDA) lead by Professor Kenner Rice has reported the synthesis and immunological evaluation for a series of haptens.
The team has emphasised the hydrolytic stability of hapten scaffolds and envisioned that hapten with amido groups at both the C3 and C6 positions should be more stable than a hapten with an ester in one of those positions. Insertion of a 3-amino group and the construction of a C-6α-(2-oxo-propyl) group stereoselectively was the challenging task towards the synthesis of designed hapten derivatives. The group has tackled the synthetic hurdle by adopting convergent synthesis approach and validating the stereochemistry by single X ray crystallography.
Furthermore, in vivo Immunological studies of all three new synthetic haptens reveals that one derivative DiAmHap (3) is chemically more stable and better candidate for a heroin vaccine than 6-PrOxyHap (1) and DiPrOxyHap (2). This work is a very positive step towards the development of vaccines for heroin abuse.
Synthesis and immunological effects of heroin vaccines
Fuying Li, Kejun Cheng, Joshua F. G. Antoline, Malliga R. Iyer, Gary R. Matyas, Oscar B. Torres, Rashmi Jalah, Zoltan Beck, Carl R. Alving, Damon A. Parrish, Jeffrey R. Deschamps, Arthur E. Jacobson and Kenner C. Rice
DOI: 10.1039/C4OB01053A