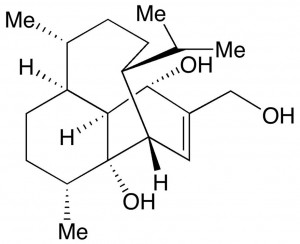
Vinigrol is a diterpenoid natural product, first isolated from the fungal strain Virgaria nigra F-5408 in 1987 by Ando and co-workers. It has been found to display biological activity against hypertension, platelet aggregation, HIV and inflammation. Despite these broad potential medicinal uses of vinigrol prompting great interest in its chemical synthesis, the first total synthesis of this molecule was not achieved until 2009, when Baran and co-workers published their outstanding stereocontrolled and fully scalable twenty three step route.1
The over two decade gap between isolation and the first published total synthesis can be attributed to the great synthetic challenges posed by the structure of vinigrol. The molecular structure of vinigrol (elucidated by X-ray crystallography) contains an unusual cis-decalin ring system, bridged by an eight membered ring, in addition to eight contiguous stereocentres. Due to this rare molecular structure, retrosynthetic analyses of vinigrol have been highly varied. A total synthesis attempt in 2003 by Paquette involved an anionic oxy-Cope rearrangement to create a decorated decalin ring system, however final closure of the eight membered ring was unsuccessful. In 2005, the same group found a ring closing metathesis strategy using Grubbs catalyst also failed to deliver the eight membered ring (Figure 2).2 Separate attempts at a vinigrol total synthesis by Fallis, Njardarson and Hanna also did not result in complete synthesis of the natural product.3 The 2009 synthesis by Baran utilized two challenging Diels-Alder reactions to create much of the cyclic core of vinigrol (Figure 2). A subsequent Grob fragmentation was the key step towards furnishing the tricyclic vinigrol core, with minimal further elaboration giving the completed natural product in 3% overall yield.
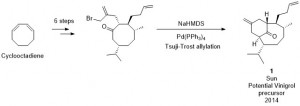
In this paper, Sun and co-workers take on this long standing challenge from a new perspective. Instead of attempting to install the troublesome eight membered ring in a late-stage transformation, they begin with an eight membered ring as a starting material, and rely on its inherent conformational bias to control the selectivity of further functionalisations. This cleverly circumvents the necessity for the ring-closing step which caused so many problems in earlier synthesis attempts. Their strategy proved successful, with two Michael additions (which indeed proved to be stereoselective, based on the natural conformation of the eight membered ring) providing three of the eight stereocentres in just two steps. Further elaboration of the ring system followed by an intramolecular Tsuji-Trost allylation reaction gave 1 (Figure 3), which, despite bearing the wrong stereochemistry at one of its six stereocentres, can be regarded as a potential late-stage precursor to the natural product vinigrol.
To read more, see;
“A novel synthetic approach to the bicyclo[5.3.1]-undecan-11-one framework of vinigrol”
Xian-Lei Wang, Yun-Yu Lu, Jie Wang, Xuan Wang, He-Quan Yao, Guo-Qiang Lin and Bing-Feng Sun,
Org. Biomol. Chem., 2014, DOI:10.1039/c4ob00046c. Free to access until 26 May
References
1 T. J. Maimone, J. Shi, S. Ashida, P. S. Baran, J. Am. Chem. Soc., 2009, 131, 17066.
2 L. A. Paquette, R. Guevel, S. Sakaoto, I. H. Kim, J. Crawford, J. Org. Chem., 2003, 68, 6096; L. A. Paquette, I. Efremov, Z. Liu, J. Org. Chem., 2005, 70, 505.
3 J. Lu, D. G. Hall, Angew. Chem. Int. Ed., 2010, 49, 2286.
Dr C. Liana Allen is currently a post-doctoral research associate in the group of Professor Scott Miller at Yale University, where she works on controlling the enantio- or regioselectivity of reactions using small peptide catalysts. Liana received her Ph.D. in organic chemistry at Bath University with Professor Jonathan Williams, where she worked on developing novel, efficient syntheses of amide bonds.