Imagine finally completing a 30 step total synthesis only to discover that the molecule you were aiming for was the wrong one. The consequences of structural misassignment of complex organic molecules can be costly and time consuming, not to mention frustrating. Now, a new NMR method aims to highlight errors in proposed structures at a much earlier stage, preventing such scenarios.
NMR spectroscopy is a standard tool for elucidating the structure of organic molecules. This may be a straightforward job when confirming the identity of small molecules. However, in the case of complex molecules, the task becomes much more difficult and errors can result in the wrong structure being proposed.
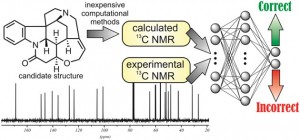
Ariel Sarotti from the Rosario National University, Argentina, has developed a new, computationally inexpensive method combining calculated and experimental 13C NMR data to flag up incorrect structures. This rapid and simple process can determine if a candidate structure is incorrect, using trained artificial neural networks (ANNs) to find patterns in both the calculated and experimental data to do the decision making. A set of 200 molecules with known correct and incorrect NMR assignments was used to create and train the system. The subsequent testing phase correctly identified the incorrect structures of a set of 26 natural products. While some knowledge of computational chemistry is required, Sarotti’s development of an Excel spreadsheet tool will allow chemists to use the method without being experts in ANNs, making it much more accessible.
Read the full story in Chemistry World
Successful combination of computationally inexpensive GIAO 13C NMR calculations and artificial neural network pattern recognition: a new strategy for simple and rapid detection of structural misassignments
DOI: 10.1039/C3OB40843D