LOC recently published a themed issue on nanomaterials and technologies for lab on a chip applications in collaboration with WAM-NANO2012 – the III International Workshop on Analytical Miniaturization and NANOtechnologies.
The conference was held last month in Barcelona, and below Arben Merkoçi provides a short summary of the meeting:
 The “III International Workshop on Analytical Miniaturization and NANOtechnologies (WAM-NANO2012)” was held in Barcelona, Spain on 11 and 12 June 2012. The event, organised by the Catalan Institute of Nanotechnology (ICN), highlighted the latest international developments in the use of nanotechnologies and nanomaterials for the design and applications of lab-on-a-chip (LOC) systems and other miniaturised analytical (bio)systems. The workshop, together with a themed issue of Lab-on-a-Chip, entitled “Nanotechnologies and nanomaterials in lab-on-a-chip”, reported on the most important achievements in analytical miniaturisation, where nanotechnology is positioned to enable great advances.
The “III International Workshop on Analytical Miniaturization and NANOtechnologies (WAM-NANO2012)” was held in Barcelona, Spain on 11 and 12 June 2012. The event, organised by the Catalan Institute of Nanotechnology (ICN), highlighted the latest international developments in the use of nanotechnologies and nanomaterials for the design and applications of lab-on-a-chip (LOC) systems and other miniaturised analytical (bio)systems. The workshop, together with a themed issue of Lab-on-a-Chip, entitled “Nanotechnologies and nanomaterials in lab-on-a-chip”, reported on the most important achievements in analytical miniaturisation, where nanotechnology is positioned to enable great advances.
WAM-NANO2012 drew 85 participants from a dozen countries and featured 19 talks by invited speakers, 42 posters, and exhibits by three companies in the field (Fluigent, MicruX Technologies and Invenios). The event began with an opening presentation by Workshop Director Prof Arben Merkoçi, who heads the Nanobioelectronics and Biosensors Group at ICN.
Invited speakers at WAM-NANO2012:
Yoshinobu Baba (Dept. Applied Chemistry, Nagoya University, Japan)
Nanobiodevice-based single biomolecule and cell analysis for cancer diagnosis and stem cell therapy
Emanuel Carrilho (Bioanalytical, Microfabrication and Separations Group, Instituto de Química de Sao Carlos/ Universidade de Sao Paulo, Instituto Nacional de Ciencia e Tecnologia de Bioanalitica, Sao Carlos Brasil)
New strategies for contactless conductivity detection in microfluidic systems and electrophoresis
Agustín Costa García (University of Oviedo, Spain)
Nanotechnological approaches for improvement of microchip electrophoresis devices
Emmanuel Delamarche (IBM Zurich Research Laboratory, Switzerland)
Miniaturized assays using capillary-driven microfluidics
Tatsuro Endo (Analytical Chemistry research Group, Department of Applied Chemistry, Osaka Prefecture University, Japan)
Printed two dimensional photonic crystal for single step label-free biosensor
Alberto Escarpa (University of Alcalà, Spain)
High NIR-purity index single-walled carbon nanotubes for electrochemical sensing in microfluidic chips
Carlos García (University of Texas at San Antonio, USA)
Microchips, robots, and nanomaterials: novel strategies for the analysis of biologically active compounds
Akihide Hibara (Institute of Industrial Science, The University of Tokyo, Tokyo, Japan)
Chemistry and physics of liquid interfaces in microfluidic devices
Alexander Kuhn (ENSCBP, University of Bordeaux, France)
Bipolar electrochemistry in microfluidic channels
Jörg P. Kutter (DTU Nanotech, Department of Micro- and Nanotechnology, Technical University of Denmark, Denmark)
Carbon nanotubes for applications in miniaturized separation systems
Laura Lechuga (Centre d’Investigacions en Nanociencia i Nanotecnologia, CIN2 (CSIC-ICN), Barcelona, Spain)
Photonic lab-on-a-chip platforms including novel bimodal interferometers, microfluidics and grating couplers
Arben Merkoçi (Institut Català de Nanotecnologia, ICN, Barcelona, Spain)
Nanoparticles based microfluidics (bio)sensing
Bradley J. Nelson (Institute of Robotics and Intelligent Systems, ETH Zurich, Switzerland)
Fabrication and magnetic actuation of polymer helical microswimmers coated with a soft magnetic film
Mar Puyol (Dept. Chemistry, Autonomous University of Barcelona, Spain)
Microfluidic platforms for nanoparticles synthesis and bioanalytical sensing
Johan Roeraade (Dept. Analytical Chemistry, Royal Institute of Technology, Sweden)
New miniaturized technologies for mass spectrometry of biomolecules
Oliver Schmidt (Leibniz Institute for Solid State and Materials Research (IFW) Dresden, Germany)
Rolled-up nanotech for lab-in-a-tube systems and microjet engines
Albert Van der Berg (University of Twente, Twente, The Netherlands)
Labs on a chip for medical applications
Jean-Louis Viovy (Macromolecules and microsystems in biology and medicine (MMBM), Institute Curie, Paris, France)
Neuron arrays on chip to study neurodegenrative diseases
Joseph Wang (Dept. Nanoengineering, University of California, San Diego (UCSD), USA)
Nanomotor-based biosensing: towards nanomachine-based lab-on-chips
View the whole issue here
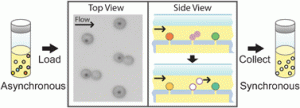
 On the outside front cover we have a HOT article from Ju Min Kim et al who have achieved viscoelasticity-driven focusing of particles as small as 200 nm – a technique which had previously only been achieved with micron-sized particles. The team also showed that DNA focusing is significantly enhanced by medium viscoelasticity and that the focussing of both the colloidal particles and DNA is dependant on length.
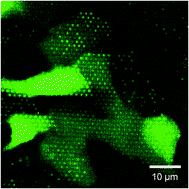
On the outside front cover we have a HOT article from Ju Min Kim et al who have achieved viscoelasticity-driven focusing of particles as small as 200 nm – a technique which had previously only been achieved with micron-sized particles. The team also showed that DNA focusing is significantly enhanced by medium viscoelasticity and that the focussing of both the colloidal particles and DNA is dependant on length. On the inside front cover is another HOT article from Noo Li Jeon and colleagues at Seoul University who have designed a 3D array of perfusable capillaries from HUVECs. The the capillary network can be grown on the microfluidic device in 3-4 days and the authors hope will not only be useful for basic angiogenesis research but also drug screening applications.
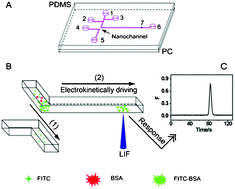
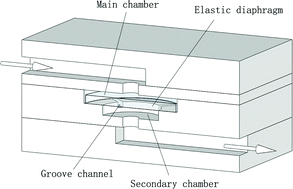
On the inside front cover is another HOT article from Noo Li Jeon and colleagues at Seoul University who have designed a 3D array of perfusable capillaries from HUVECs. The the capillary network can be grown on the microfluidic device in 3-4 days and the authors hope will not only be useful for basic angiogenesis research but also drug screening applications. Last but not least, on the back cover is research from Anup Singh and colleagues at Sandia National Laboratory who have developed a chip that enables dynamic monitoring of an entire cell signalling pathway in a single experiment, by combining cell culture, stimulation, and preparation for analysis by multicolor flow cytometry and fluorescence imaging in one device.
Last but not least, on the back cover is research from Anup Singh and colleagues at Sandia National Laboratory who have developed a chip that enables dynamic monitoring of an entire cell signalling pathway in a single experiment, by combining cell culture, stimulation, and preparation for analysis by multicolor flow cytometry and fluorescence imaging in one device.