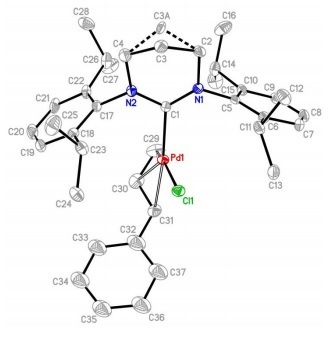
N-heterocyclic carbenes are neutral σ donors and electron rich, making them attractive for use as ligands in transition metal catalysts. By changing the nature of the azole ring (nitrogen heterocycle containing at least one other atom of S, N or O) and the substituents, the electronic and steric properties can be modified.
Studies of expanded NHCs containing a 6- or 7-membered ring have only recently started to appear in the literature. This includes that of Nechaev and colleagues who have synthesized a series of new 6- and 7-membered NHC palladium complexes. Kolychev and Nechaev screened the complexes as catalysts for Suzuki-Miyaura cross-coupling of heteroaryl chlorides and bromides. They tested the catalysts using ‘green‘ reaction conditions and showed that reaction products are clean with no homo coupling products formed.
These systems are very attractive – it will be exciting to see how expanded ring NHCs are utilized more in the future…
Download the full Dalton Transactions paper to find out more:
Expanded Ring Diaminocarbene Palladium Complexes: Synthesis, Structure, and Suzuki–Miyaura Cross-Coupling of Heteroaryl Chlorides in Water
Eugene Kolychev and Mikhail Nechaev
Dalton Trans., 2013, DOI: 10.1039/C3DT32860K
Expanded ring N-heterocyclic carbenes: A comparative study of ring size in palladium (0) catalysed Mizoroki–Heck coupling