Iron is a biological necessity and the amount we take-in must be carefully controlled – normally this isn’t a problem as eating a varied, healthy diet provides us with the nutrients we need. Once we have an adequate concentration of iron our intestines then stop absorbing it, preventing a surplus building up.
This absorption control is usually very effective as eating and drinking is our main source of iron, however after a blood transfusion excess iron can be introduced into the body and there is no natural mechanism to remove the unwanted metal ions. Some diseases such as Diamond–Blackfan anemia or β-thalassemia can require regular treatment by blood transfusion, which can lead to acute iron overload. This can be very harmful as iron can build up in vital organs such as the heart and liver and cause irreversible organ damage.
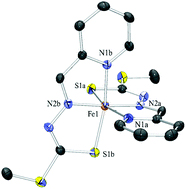
Iron chelating agents offer a treatment for Fe overload by binding to the excess iron ions, allowing them to be safely excreted before they build up and cause damage to the body. This Hot Article by Professor Des Richardson and Professor Paul Bernhardt et al. details the synthesis and characterisation of a series of dithiocarbazate Schiff base analogues for their suitability as iron chelators. Schiff bases are previously known as chelation ligands, but the derivatives detailed here show some interesting biological activities…
l
Heterocyclic dithiocarbazate iron chelators: Fe coordination chemistry and biological activity
Maram T. Basha, Jy D. Chartres, Namfon Pantarat, Mohammad Akbar Ali, Aminul Huq Mirza, Danuta S. Kalinowski, Des R. Richardson and Paul V. Bernhardt
Dalton Trans., 2012, Advance Article
DOI: 10.1039/C2DT12387H
This article will be part of our upcoming themed issue on Metals in Medicine: Non-Cancer Therapeutics – other hot papers due to be published in this special themed issue include the HOT Article: Anti-microbials that target destabilised DNA and the Hot Perspective: Treating Wilsons Disease.
You can also keep up to date with the latest news in inorganic chemistry by following us on twitter or signing up to our e-alert service.