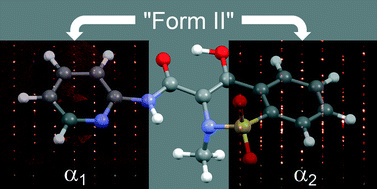
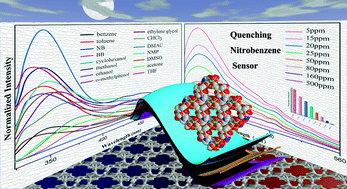
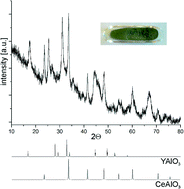
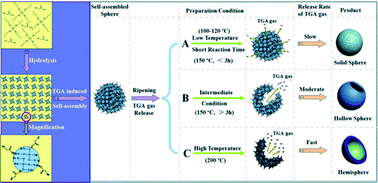
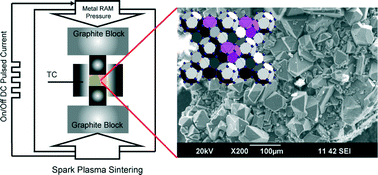
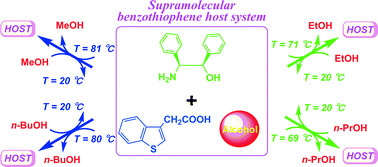
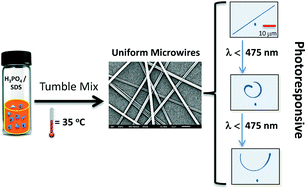
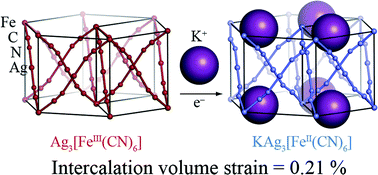
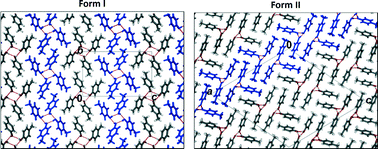
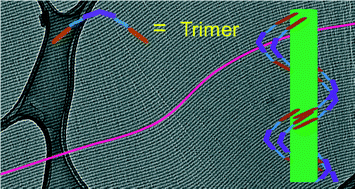
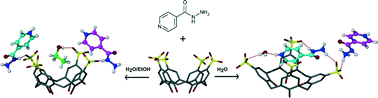
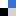The 3rd China-India-Singapore (CIS) Symposium on Crystal Engineering was recently held at the Indian Institute of Science, Bangalore, India, 8–10 December, 2014.
The CIS symposium series on “Crystal Engineering” was conceived so that researchers from China, India and Singapore could meet, discuss, present and exchange their research work to accelerate the growth of crystal engineering.
The symposium series aims to discuss and acknowledge recent advances in the field of crystal engineering in the south Asian region. The 1st CIS Symposium on Crystal Engineering was held at National University of Singapore (NUS) from 30 July –
2 August, 2010. The 2nd CIS Symposium on Crystal Engineering was held at Guangzhou, China, 20–23 November, 2012.
The 3rd CIS Symposium on Crystal Engineering was recently organized by Professors Gautam. R. Desiraju and S. Natarajan at the Solid State and Structural Chemistry Unit, at the Indian Institute of Science. A total of 22 researchers from different institutes in China, India and Singapore presented their work, ranging from the research areas of porous materials and pharmaceutical solids to intermolecular interactions and computational chemistry. This symposium highlighted some of the recent developments in the field of organic, inorganic, pharmaceutical and organic-semiconductor materials achieved through the applications of crystal engineering.

The 3rd CIS Symposium on Crystal Engineering started with the welcome address from Prof. S. Natarajan (IISc, India) followed by inaugural speeches by Prof. Gautam R. Desiraju (IISc, India) and Prof. Shilun Qiu (Jilin University, China). This was followed by the first scientific lecture by Prof. X.M. Chen (Sun Yat-Sen University, China) on “Metal Organic Frameworks for Molecular Oxygen Sensing”. Prof. T.N. Guru Row (IISc Bangalore, India), Prof. A. Ramanan (IIT Delhi, India), and Prof. Cheng Peng (Nankai University, China) described the use of the crystal engineering concepts towards understanding the a) relevance of intermolecular interactions, b) structure and properties of metal carbarboxylate-based supramolecular assemblies and c) rational design of molecular magnetic materials. Other speakers of the first day were Prof. S. Aipitamula (A*STAR, Singapore), Prof. P. Dastidar (IACS, India), Prof. C. Malla Reddy (IISER Kolkata, India), Prof. P. Thilagar (IISc Bangalore, India), and Prof. P. Venugopalan (Punjab University, India).
On the second day of the meeting, Prof. J.J. Vittal (National University of Singapore, Singapore) outlined an interesting project on “Crystal Engineering of Photoreactive and Photosalient Crystals”. This was followed by the scientific lectures by Profs. Lu Tong-Bu (Sun Yet-Sen University, China), R. Banerjee (CSIR-NCL, India), V.R. Pedireddi (IIT Bhubaneswar, India), P.S. Mukherjee (IISc Bangalore, India), Su Cheng-Yong (Sun Yet-Sen University, China), J.N. Moorthy (IIT Kanpur, India), T.K. Maji (JNCASR, Bangalore, India), A. Nangia (University of Hyderabad, India), B. K. Saha (Pondicherry University, India) and Daliang Zhang (Jilin University, China).
Prof. Shilun Qiu, (Jilin University, China) delivered a stimulating talk on gas storage in metal-organic frameworks and covalent-organic frameworks during the final day of the meeting. His talk was followed by the scientific lectures by Profs. K. Biradha (IIT Kharagpur, India), and T.S Thakur (Central Drug Research Institute, India).
The next meeting will be held in 2016 as a South and East Asia Conference on Crystal Engineering, SEACCE. This meeting will aim to popularize the idea of crystal engineering in countries such as Bangladesh, Malaysia, Nepal, Pakistan, Sri Lanka, Thailand, and Vietnam. A resolution was signed by the participants from China, India and Singapore. The next meeting is likely to be held in Nepal, Sri Lanka or Bhutan in the summer of 2016.
Comments Off on Conference Report: 3rd China-India-Singapore Symposium on Crystal Engineering