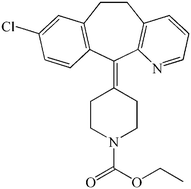
Drugs are often prepared in crystalline form. However, the relative thermodynamic stability of crystalline compounds can limit water solubility and bioavailability in the body. An alternative is to make the drugs in an amorphous form that is less stable and more soluble. Unfortunately, this form tends to revert to the crystalline form during manufacture, processing or storage, which can limit its usefulness. A new paper by Aiguo Zeng et al., aims to increase understanding of the crystallisation of amorphous drugs, using loratadine (shown below) as a model compound.
Loratadine is a widely-available, non-sedating anti-histamine, used to treat hay-fever and other allergies. It has been on the market since 1993 and is included in the World Health Organisation’s Model List of Essential Medicines.
Four amorphous samples were prepared by quench-cooling – where molten compounds are rapidly cooled so they have insufficient time to arrange into a crystal lattice. Study of the samples obtained by cooling at 298 K, 277 K, 253 K and 233 K showed no significant differences in the crystallisation mechanism with quenching temperature.
However, the tendency to crystallise increased with decreasing quench-cooling temperature. Authors attribute this to the differences in molecular mobility and relaxation of the samples, especially the so-called Johari-Goldstein process of loratadine, involving motion of all atoms in the molecule. This finding will allow the preparation of loratadine with a lower tendency to crystallise but which retains the beneficial properties of the amorphous form. It will also inform studies of the amorphous form of other drug molecules.
For more details, read the full paper at:
Ruimiao Chang, Qiang Fu, Yong Li, Mingchan Wang, Wei Du, Chun Chang and Aiguo Zeng
CrystEngComm, 2017, Advance Article
DOI: 10.1039/C6CE01645F, Paper
_______________________________________________________________________________
 Gwenda Kyd has a PhD in metallocarborane chemistry from the University of Edinburgh. Other research work includes the spectroscopic study of the structure of glasses and organometallic electron-transfer reactions and the preparation of new inorganic phosphors. She published a book, ‘Molecules, Medicines and Mischief’, in 2014, on some of the chemicals found in plants and is currently working on a follow-up.
Gwenda Kyd has a PhD in metallocarborane chemistry from the University of Edinburgh. Other research work includes the spectroscopic study of the structure of glasses and organometallic electron-transfer reactions and the preparation of new inorganic phosphors. She published a book, ‘Molecules, Medicines and Mischief’, in 2014, on some of the chemicals found in plants and is currently working on a follow-up.