Although big strides have been made in the development of anti-cancer drugs, a major hindrance in their development is the lack of appropriate models to test their efficacy. 2D models are poor representations for real-life situations, while animal models present ethical issues. 3D cell-based tumour models would be a major improvement, yet transport modes in 3D models are poorly understood, impeding targeted strategies. A new study published in Biomaterials Science by Lu et al. elucidates how polymeric micelles, a main cancer-treatment platform, are taken up and transported, allowing for improved development of strategies to effectively deliver drug payloads to tumours.
Despite the plethora of anti-cancer drugs developed in the past decades, testing efficacy prior to human trials remains suboptimal at best. 2D cell models poorly reflect the real-life situation, with different pharmacokinetics and nutrient availability, leading to misleading observations and faulty conclusions. Better representations are animal tumour models, yet – separate from obvious ethical concerns – animal metabolism is not necessarily comparable to humans. The development of a more representative 3D multicellular tumour spheroid (MCTS) model to investigate treatment modalities would be a big step towards alleviating those issues, yet the mode of drug carrier transport in 3D models is poorly understood, impeding targeted strategies.
To address the current caveats in MCTS knowledge, Dr. Martina Stenzel’s research group at the University of New South Wales investigated how polymeric micelles are taken up and transported through the outer layers of MCTS’s. Her group created both crosslinked and uncrosslinked polymeric micelles (called CKM and UCM, respectively) and delivered them to a pancreatic MCTS’s. The penetration was monitored using a fluorescent payload, Nile Red, and various modes of endocytosis, as well as exocytosis, were blocked. Their results indicate that it is both caveolae-mediated endocytosis and exocytosis mechanisms are required for good penetration depth of micelles into MCTS’s. Taken together, this evidence points toward transcellular mechanisms as the primary mode of transport for drug-loaded polymeric micelles.
Dr. Stenzel’s group further shows that UCM micelles could not penetrate as far as CKM micelles. The rapid release of their toxic payload doxorubicin creates an apoptotic peripheral cell layer, leading to cessation of additional transcellular transport. Perhaps the most captivating aspect of her research, though understated in the main article, is that the mode of micellar transport seems to be identical in other tumour models.
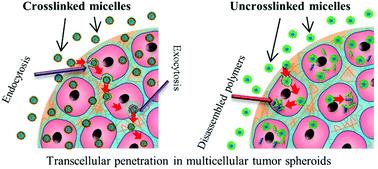
The primary mechanism for micellar penetration in MCTS models shown in this article creates important guidance to other researchers investigating anti-cancer drug delivery to tumours. An essential insight is that micelles need to be capable of retaining their structural integrity long enough to prevent payload-induced penetration limitations. Intriguingly, there are indications that the shown penetration mechanisms are extrapolatable to other tumours as well. This study therefore represents a great step forward towards creating better utilization of in vitro tumour models.
Check out the full article:
H. Lu, R.H. Utama, U. Kitiyotsawat, K. Babiuch, Y. Jiang and M.H. Stenzel
Biomater. Sci., 2014, Advance Article, DOI: 10.1039/C4BM00323C

Robert van Lith (@RvLith) is currently a Post-Doc in the Biomedical Engineering department at Northwestern University, developing intrinsically antioxidant biomaterials. He recently received his Ph.D. from Northwestern University for his work on citrate-based antioxidant polyesters, receiving an American Heart Association Fellowship and Society for Biomaterials award for his work. He was trained in the Netherlands, holding an M.S. degree in Biomedical Engineering from Eindhoven University of Technology. Read more about Robert’s research publications here.
Follow the latest journal news on Twitter @BioMaterSci or go to our Facebook page.