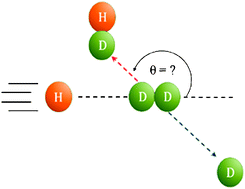
Scientists have pinpointed the moment that the CO bond, the strongest bond of any diatomic molecule, breaks when oxidised by a gold catalyst.
 Until now, research has only focussed on the catalysts’ active site and not on the reaction mechanisms. Graham Hutchings, Albert Carley and colleagues at the University of Cardiff have investigated the reaction mechanism occurring on a Au/Fe2O3 catalyst and found that CO dissociates at ambient temperature when co-adsorbed with O2.
Until now, research has only focussed on the catalysts’ active site and not on the reaction mechanisms. Graham Hutchings, Albert Carley and colleagues at the University of Cardiff have investigated the reaction mechanism occurring on a Au/Fe2O3 catalyst and found that CO dissociates at ambient temperature when co-adsorbed with O2.
‘The oxidation of CO gives a surprising result on a gold catalyst as the CO bond being broken is counter-intuitive since it is the strongest diatomic bond,’ says Hutchings.
Read the rest of the Chemistry World article…
View the PCCP paper:
CO bond cleavage on supported nano-gold during low temperature oxidation
Albert F. Carley, David J. Morgan, Nianxue Song, M. Wyn Roberts, Stuart H. Taylor, Jonathan K. Bartley, David J. Willock, Kara L. Howard and Graham J. Hutchings
Phys. Chem. Chem. Phys., 2011, DOI: 10.1039/c0cp01852j










 Read the
Read the  Until now, research has only focussed on the catalysts’ active site and not on the reaction mechanisms. Graham Hutchings, Albert Carley and colleagues at the University of Cardiff have investigated the reaction mechanism occurring on a Au/Fe2O3 catalyst and found that CO dissociates at ambient temperature when co-adsorbed with O2.
Until now, research has only focussed on the catalysts’ active site and not on the reaction mechanisms. Graham Hutchings, Albert Carley and colleagues at the University of Cardiff have investigated the reaction mechanism occurring on a Au/Fe2O3 catalyst and found that CO dissociates at ambient temperature when co-adsorbed with O2. ‘
‘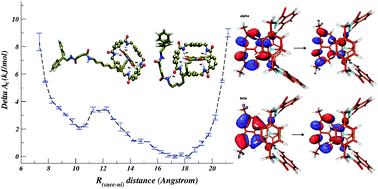 Molecular dynamics simulations and DFT calculations support experimental observations providing some insights into mechanical and electronic properties of a molecular shuttle working in acetonitrile solution.
Molecular dynamics simulations and DFT calculations support experimental observations providing some insights into mechanical and electronic properties of a molecular shuttle working in acetonitrile solution.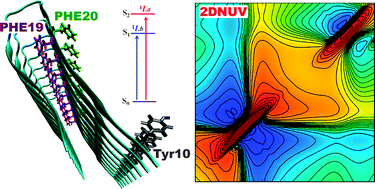 ‘HOT’ article
‘HOT’ article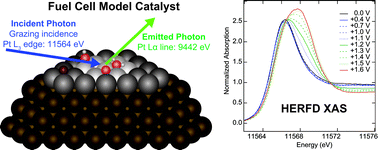 View the article on PhysOrg.com
View the article on PhysOrg.com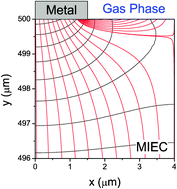
 Three novel inorganic–organic framework compounds containing different organic chromophore ligands have been synthesized and evaluated for their use as phosphor materials for solid state lighting and other applications.
Three novel inorganic–organic framework compounds containing different organic chromophore ligands have been synthesized and evaluated for their use as phosphor materials for solid state lighting and other applications.