 We invite you to join us for a joint PCCP–UCL symposium on Tuesday 15th October 2013.
We invite you to join us for a joint PCCP–UCL symposium on Tuesday 15th October 2013.
Following a welcome by Professor Helen Fielding and brief introduction of PCCP by Editor Philip Earis, the following international speakers, including members of the journal’s Editorial Board, will talk about some of their exciting research:
14:10
Katsuhiko Ariga, NIMS, Japan
Manual nanotechnology: a new page in the nanotechnology manual
14:40
Cyrus Hirjibehedin, London Centre for Nanotechnology
Tunable single molecule magnetoresistance driven by magnetically sensitive negative differential resistance
15:10
Gaoquan Shi, Tsinghua University, China
Graphene based electrochemical devices
15:50
Daniela Goldfarb, Weizmann Institute of Science, Israel
Gd3+ spin labeling for structural interrogation of biomolecules
16:20
David Rueda, Imperial College London
Watching AID scan ssDNA and transcribed dsDNA with single molecule resolution
Location: Chemistry Lecture Theatre, Department of Chemistry, UCL, 20 Gordon Street, London WC1H 0AJ.
No registration necessary.
**** The lectures will be followed with a wine reception ****






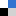




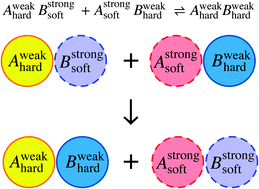 When I was perusing the ‘recently published’ pages for something to write about, I was moderately surprised to stumble on this article. It immediately caught my eye as being unlike anything I’d ever seen in
When I was perusing the ‘recently published’ pages for something to write about, I was moderately surprised to stumble on this article. It immediately caught my eye as being unlike anything I’d ever seen in 
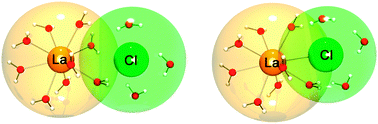
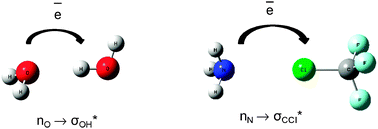
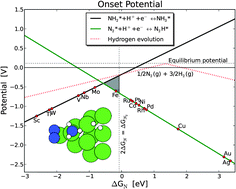
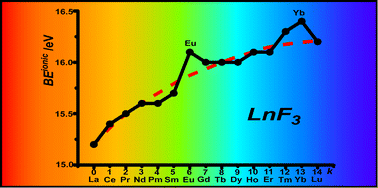 The complex electronic structure of the lanthanides and actinides is largely a mystery. The f-shell is not readily explained in our theoretical treatment of atoms and molecules, and coupled with the difficulty of obtaining readily interpreted experimental data. The result is that we are still struggling to understand the elements at the bottom of the periodic table. Xu and co-workers conduct a good and meticulous computational study of lanthanide trifluorides, and do significantly increase our understanding of the rare earth elements, albeit in simple molecules in the gas phase. My problem is that I work in aqueous solution, where lanthanide ions have not just three ligands—as the molecules investigated in this paper have—they have eight, nine or ten.
The complex electronic structure of the lanthanides and actinides is largely a mystery. The f-shell is not readily explained in our theoretical treatment of atoms and molecules, and coupled with the difficulty of obtaining readily interpreted experimental data. The result is that we are still struggling to understand the elements at the bottom of the periodic table. Xu and co-workers conduct a good and meticulous computational study of lanthanide trifluorides, and do significantly increase our understanding of the rare earth elements, albeit in simple molecules in the gas phase. My problem is that I work in aqueous solution, where lanthanide ions have not just three ligands—as the molecules investigated in this paper have—they have eight, nine or ten.