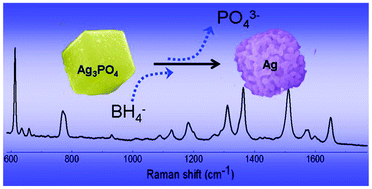
In this HOT article, Shengchun Yang and co-workers describe the preparation of porous Ag nanocubes by synthesizing Ag3PO4 nanocubes and using them as hard templates. With the addition of strong reducing agents, Ag is reduced in situ to form porous Ag. The resulting Ag showed good SERS (surface enhanced Raman scattering) properties by detecting nanomolar concentrations of Rhodamine 6G.
Volume shrinkage induced formation of porous Ag sub-microcubes via solid–liquid reaction for SERS
Find out more by downloading the full paper now:
Volume shrinkage induced formation of porous Ag sub-microcubes via solid–liquid reaction for SERS
Xiao Luo, Shaomin Lian, Liqun Wang, Shengchun Yang, Zhimao Yang, Bingjun Ding and Xiaoping Song
CrystEngComm, 2013, Advance Article
DOI: 10.1039/C3CE27089K
You may also be interested in other publications concerning SERS and noble metals from recent studies by the same author:
A new route for the synthesis of polyhedral gold mesocages and shape effect in single-particle surface-enhanced Raman spectroscopy
Jixiang Fang, Sergei Lebedkin, Shengchun Yang and Horst Hahn
Chem. Commun., 2011, 47, 5157-5159
DOI: 10.1039/C1CC10328H
Interface synthesis of gold mesocrystals with highly roughened surfaces for surface-enhanced Raman spectroscopy
Hongjun You, Yuetian Ji, Liang Wang, Shengchun Yang, Zhimao Yang, Jixiang Fang, Xiaoping Song and Bingjun Ding
J. Mater. Chem., 2012, 22, 1998-2006
DOI: 10.1039/C1JM13211C, Paper
Also, keep up to date and follow us on twitter: @crystengcomm