Have you seen the video cover from CrystEngComm?
Have you seen the video cover from CrystEngComm?
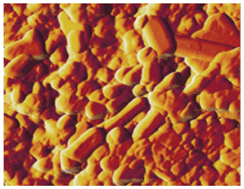
Illustrating the work published in their CrystEngComm article, Bergström et al. provide us with an overview of the growth process of self-assembled mesocrystals and ordered arrays from iron oxide nanocubes. By using visible light video microscopy, the authors were able to follow the movement of the growth crystal growth front over several minutes, capturing the transition from symmetrical “coffee-ring” deposition to dendritic or “finger-like” growth. The article forms part of the ‘Nanocrystal growth via oriented attachment’ themed issue which is available here.
In a first for both CrystEngComm and the Royal Society of Chemistry, a short video clip of the crystal growth has been embedded into the journal cover which you can access online here. To view the video, simply click on the main cover image and watch the cover come to life!* Adobe Acrobat 9 or higher is required (http://get.adobe.com/reader/). If the video does not play in your browser, try downloading it and opening it in Adobe Acrobat on your desktop.
We’re on the look-out for innovative ways to showcase research so do not hesitate to get in touch if you have a video or animated graphics to help tell the story of your published CrystEngComm article.
We’ve been experimenting with some other engaging ways of adding the authors’ video to both the cover and the article.
Video on the article landing page
We’ve added the full version of the video to the article landing page here, where it appears below the traditional abstract text and image. To enable us to embed the video we uploaded it to our YouTube channel, RSC Journals, which has the added benefit of making it available to people looking for chemistry videos on YouTube.
Video on your tablet or smartphone
You can also watch the full video on your tablet or smartphone using a free augmented reality app called Junaio Augmented Reality Browser. To do this you’ll need a tablet or phone that’s connected to the internet and has a camera on the back.
To watch the video:
• Use you app store of choice to install Junaio.
• Get the cover image ready to scan. You can open the article pdf from here to the first page on your computer screen, print the first page (this will work for both colour and greyscale printing), or just scan the image in this blog post.
• Open the Junaio app, position your phone or tablet so that the cover image is on the screen, and press the scan button in the top right corner of the screen.
• The Royal Society of Chemistry logo will appear briefly in the top left corner, indicating that you have connected to our channel, and then the YouTube video page will be displayed.
• Press play and enjoy the video.
One of the nice things about the augmented reality version is that the video is associated with the image, not just the cover, which makes it quite versatile. For example, the authors could use the image (without the play button and the journal branding) on a conference poster and people with a suitable phone or tablet would also be able to scan it and watch the video.
Please do have a look and let us know what you think in the comments. Which versions are good and which could be better? What are your favourite features and why? Is there anything else you would like to see?
For more information about the science behind the video, read the free-to-access CrystEngComm article using the link below and watch the video on the article landing page.
Dynamic growth modes of ordered arrays and mesocrystals during drop-casting of iron oxide nanocubes
CrystEngComm, 2014, 16, 1443-1450
Please do let us know what you think by commenting below! Would you like to see more videos in Royal Society of Chemistry journals?