Potentiometry to study the reversible redox chemistry of polyoxometalates.
In this Hot article, Alisa Rudnitskaya and co-workers (Aveiro University, Portugal) report on the use of potentiometric chemical sensors for the study of redox reactions of vanadium containing Keggin-type polyoxomolybdates, H5[PVV2Mo10O40]11H2O and K4[PVVMo11O40]6H2O.
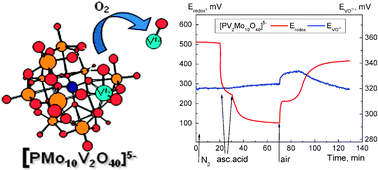
Variations in the concentrations of vanadyl ions (VO2+) during the redox reactions of POMs were followed in the real-time. Apparently, release of vanadyl from the coordination sphere of reduced di-substituted POM occurred as the response to the presence of molecular oxygen. The use of potentiometry for this purpose is innovative, and the work provides an additional perspective in ongoing efforts to understand the reversible redox chemistry of these complexes when used in catalytic aerobic oxidation.
Studies on the redox turnover of polyoxometalates using potentiometric chemical sensors by Alisa Rudnitskaya, Jose A. F. Gamelas, Dmitry V. Evtuguin and Andrey Legin; New J. Chem., 2012, Advance Article; DOI: 10.1039/C2NJ21053C, Paper
Sensing properties of NiO-doped SnO2 polyhedra
The next Hot article provided by Chinese researchers (Z. Lou, L. Wang, T. Fei and T. Zhang, Jilin University) describes the preparation, characterisations and application novel NiO-doped SnO2 polyhedra.
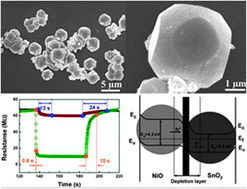
According to the authors, this hydrothermal synthesis of uniform NiO-doped SnO2 polyhedra is a facile, mild, and low-cost method to fabricate micro- and nanostructures. The application of the material in the field of resistive sensors for the revelation of ethanol has also been proposed: the main result is an enhancement of the response and recovery times in comparison with pure SnO2 based sensors. The effect of NiO doping on the response and response–recovery time towards ethanol is also discussed.
Enhanced ethanol sensing properties of NiO-doped SnO2 polyhedra by Zheng Lou, Lili Wang, Teng Fei and Tong Zhang, New J. Chem., 2012, Advance Article, DOI: 10.1039/C2NJ21030D, Paper
Interested to know more? Why not read by accessing the full articles now and let us know your thoughts and comments below! These “Hot articles” will be free to access for a period of 4 weeks.
To stay up-to-date with the latest NJC developments, sign up to its free table-of-contents email alert!