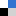Supramolecular self-assembly can be described as a spontaneous process of association of individual molecules to construct complex and large architectures with distinct features from the corresponding monomers. Encouraged by the natural self-assembly processes, creating new artificial self-assembled systems with desired properties has become a pressing research area of interest. Accordingly, during the last few decades, the scientific community has invested enormous effort in searching and constructing novel self-organized organic architectures with diverse functionalities, which have registered their potential in a wide range of applications, such as biosensing, drug delivery, tissue engineering, organic electronics, catalysis, and others.
During the last few decades, pyrene, a small polyaromatic hydrocarbon, has captured an immense attention to the scientific community as a unique blue emissive fluorophore. Pyrene has been an outstanding choice in organic functional materials because of its superb photophysical characteristics and versatile applications, and the possibility of synthetic modifications on the pyrene core. In comparison to systems that only enable self-assembly, the examples of pyrene-based transient assembly and further disassembly remain elusive in the past literature. Transient assembly is an out-of-equilibrium process which is thermodynamically unstable, and hence it dissociates into monomers via the disassembly process.
In the search of such assembly–disassembly system, the group of Prof. Apurba Lal Koner of the IISER Bhopal, India, in collaboration with Prof. Anup Pramanik at Sidho-Kanho-Birsha University, have developed a new biogenic amine (BA)-induced pyrene-based transient assembly and spontaneous disassembly system to access blue emissive Py-BA conjugated monomers which exhibited solid-state emissive property in addition to lysosomal targeting application (Figure 1).
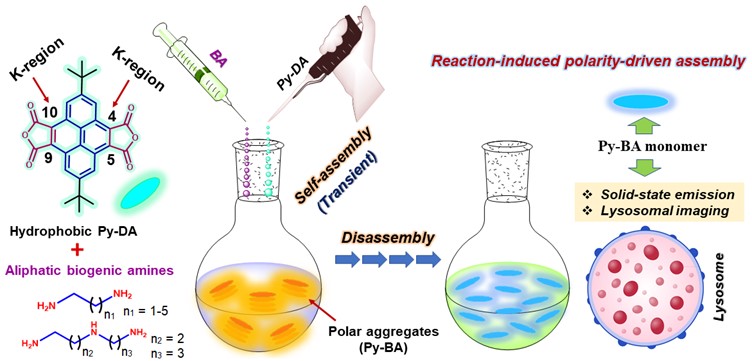
Figure 1. Representation of reaction-based transient assembly and disassembly and demonstration of solid-state emission and bioimaging application of the monomers.
The group has demonstrated that the nucleophilic reaction between BA and pyrene-anhydride led to form open polar conjugates which created the transiently assembled network driven by strong π–π and multiple H-bonding interactions. The evolution of transient assembly via ground-state pre-association was fully established by a combination of spectroscopic and microscopic techniques in addition to computational study. Addition of BA such as 1,4-DAB in probe solution governed to generate an intense excimer band near 550 nm (yellow emission) in fluorescence spectra which further showed quenching with BA concentration as well as time (Figure 2a–b).
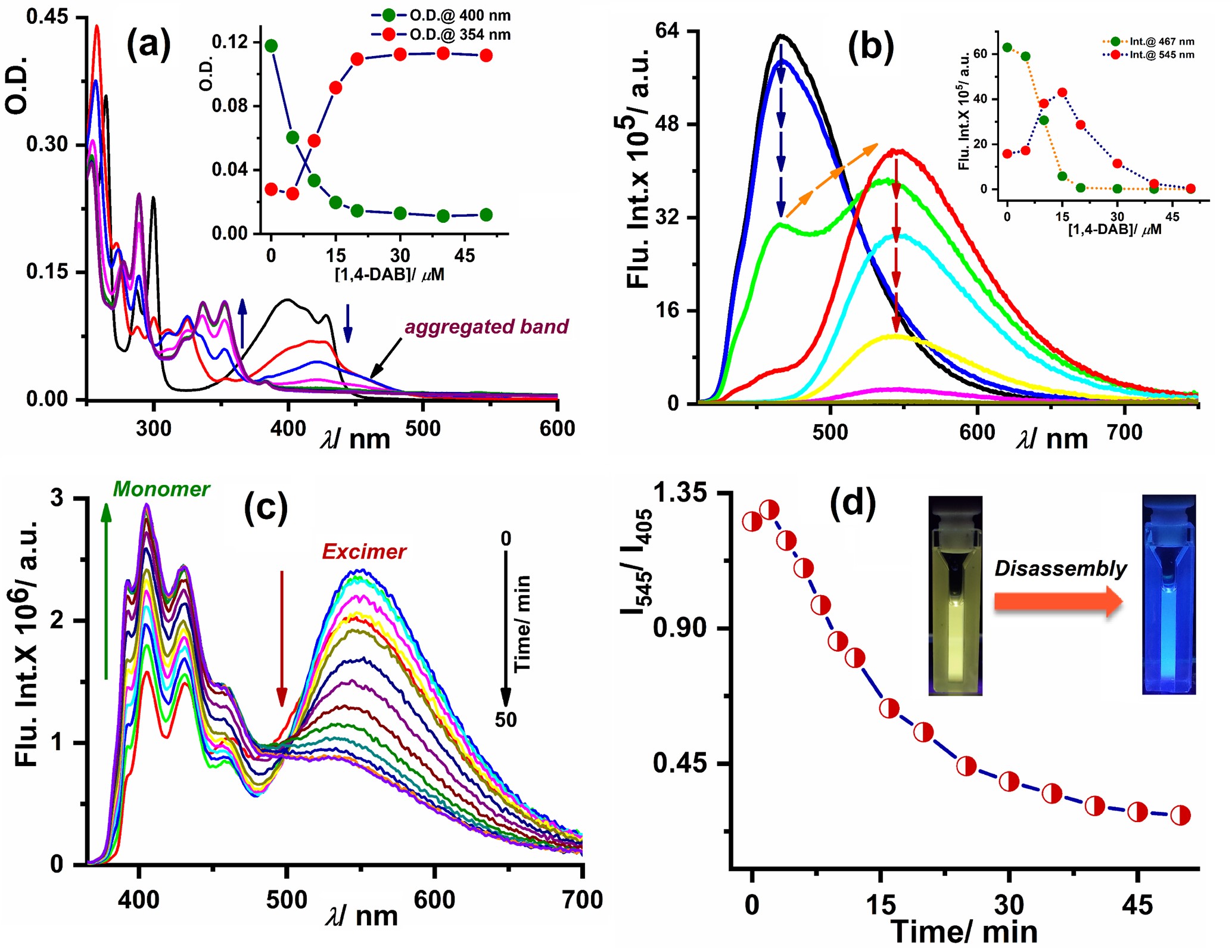
Figure 2. (a) UV-Vis. and (b) fluorescence spectra of Py-DA (5 µM) with increasing concentration of 1,4-DAB (0–50 µM), inset showing the change of O.D. and fluorescence intensity with concentration. (c) representation of temporal intensity changes in excimer and monomer band in CHCl3. (d) plot of change of I545/ I405 ratio with time; inset cuvette images showing transformation of fluorescence color from excimer to monomer species.
Moreover, time-dependent quenching of excimer band with concomitant enhancement of monomer emission near 400 nm signified dissociation of yellow-emissive excimer/ aggregation to blue-fluorescent monomers in solution (Figure 2c–d). SEM study for Py-1,4-DAB conjugate unravelled temporal evolution of larger bulk aggregates at initial time which were transformed to interconnected network morphology due to disassembly (Figure 3a–c). Two types of products, i.e., Py-BA monomers and dimer were mainly responsible for demonstrating such assembly–disassembly phenomenon in solution as evident from NMR and mass spectrometry. The blue-emissive monomer conjugates exhibited yellowish-orange emission in solid-state (Figure 3d) which can be highly useful for fabricating light-emitting devices in the future. Interestingly, the newly formed water-soluble selective Py-BA conjugates were found to be of potential significance as lysosome targeting fluorophores (Figure 3e–f). Their lysosomal staining property provided a great advantage to monitor the lysosomal membrane degradation in a time-dependent manner.
Figure 3. (a)–(c) Time-dependent SEM images of Py-1,4-DAB conjugate in CHCl3 showing morphological change with time. (d) CIE-coordinate of Py-1,4-DAB solid-state emitter; inset shows fluorescence image of thin-film captured upon illuminating at 365 nm. CLSM images of live BHK-21 cells: (e) merge image of Py-Spermine and lysotracker red (f) scatter plot with Pearson’s correlation coefficient 0.96 ± 0.02 (scale bars: 20 µm).
The work represents an intriguing example of harvesting water-soluble fluorophores with multifunctional features from reaction-induced transient assembly–disassembly processes.
Read the full article in Materials Chemistry Frontiers:
Apurba Lal Koner is an associate professor in the Department of Chemistry at the Indian Institute of Science Education and Research Bhopal (India). He received his PhD in Chemistry from Jacobs University Bremen (Germany) in 2009. From 2009 to 2012 he did postdoctoral studies at the University of Oxford (UK). Professor Apurba Lal Koner’s research team is working at the interface of chemistry, biology, and emissive materials and their applications in various research domains. He is the author of more than 95 articles of international repute and cited more than 2600 times with an index H = 25.
Web Page link: https://bionanolab.wixsite.com/akoner-iiserb