Greiner, A.1,2, Tekwa, E.W.1,3, Gonzalez, A.1, Nguyen, D.4
1Department of Biology, McGill University, 1205 Dr. Penfield, Montreal, QC, H3A 1B1, Canada.
2Department of Ecology and Evolution, University of Toronto, 25 Willcocks Street, Toronto, ON, M5S 3B2, Canada
3Department of Ecology, Evolution, and Natural Resources, Rutgers University, 14 College Farm Road, New Brunswick, NJ, 08901, USA.
4Meakins Christie Laboratories, Research Institute of the McGill University Health Centre, and Department of Medicine, McGill University, 1001 Decarie Blvd, Montreal, QC, H4A 3J1, Canada.
Why is this useful?
Microfluidic devices are used for many different types of experiments across the medical, ecological and evolutionary disciplines (Park et al., 2003; Keymer et al., 2008; Connell et al., 2013; Hol & Dekker, 2014). For example, microfluidic devices for microbial experiments require inoculation into smaller chambers that simulate natural microbial environments such as porous soils (Or et al., 2007) and biological hosts (Folkesson et al., 2012). These devices often involve complicated pump setups and irreversible seals. We developed a technique that requires only common lab equipment and makes the device reusable while also allowing the microbes to grow undisturbed (based on Tekwa et al., 2015; Tekwa et al., in review). Here, we provide a detailed guide for the assembly and the previously undocumented non-destructive disassembly of polydimethylsiloxane (PDMS) experimental devices to recover microbes in situ, which can then be plated for relative counts and further molecular analyses of population changes. This is complemented by videos for each step.
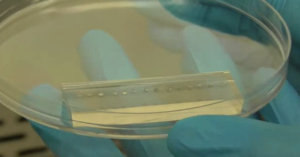
Figure 1: Microfluidic device containing 14 habitats on an elastomer (PDMS) layer pressed onto a 60mm x 24mm glass cover slip. The habitats are 10 or 20µm in depth, range from 1400 µm to 2670 µm in diameter and take the shape of a ring or network of patches. This device is used to test the effects of habitat patchiness on microbe dynamics. Habitats were dyed blue for visualization. For more information see Tekwa et al. (2015).
What do I need?
- Single-layer PDMS devices with habitats on one side
- Pipette + sterile pipette tips
- Sterile petri dishes (1/device)
- Sterile tweezers
- Inoculum
- Filtered water
- Kimwipes
- Autoclavable plastic container
- Ethanol
- Tinfoil
- Sterile 1μL inoculating loop (1/habitat)
- Sterile eppendorfs
- Phosphate-buffered saline (PBS)
- Biological safety cabinet (BSC)
How do I do it?
- Clean the PDMS devices: The PDMS device should be pre-treated once with 0.01N HCl for one hour and plasma-treated to keep it hydrophilic and amenable to bonding to glass or plastic substrates (Cho et al., 2007; Tekwa et al., 2015). Fill autoclavable plastic container 1/3rd full of 70% ethanol and PDMS devices and then cover with tinfoil, let them sit in this for >30 minutes before carefully disposing of the ethanol down the sink. Fill and empty the container with water 10 times in order to rinse the devices. Lastly, fill the container with filtered water, seal with tin foil and autoclave in order to sterilize the devices.
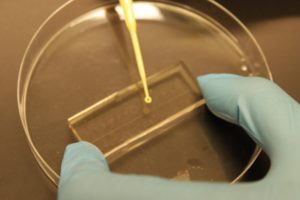
- Inoculating the devices (perform in a Biological Safety Cabinet, BSC): Set the devices to dry in the BSC for 30 minutes. Place the devices with features facing up on the lid of a petri dish and place a small amount (i.e. 0.7 µL) of inoculum onto each (of 14 in sample device Fig. 1) habitat. The amount of liquid must be sufficient to fill the habitats, but not too much as to prevent bonding between PDMS and the cover glass/petri dish (Fig. 2). Using sterile tweezers, pick up the device and place face down into the centre of a petri dish or cover glass, sealing it to the surface by pushing on the back with gloved fingers repeatedly, using a kimwipe to wick excess liquid away from the side. Then surround, but not touch, the device with kimwipes soaked in filtered water (Fig. 3) to ensure that the device does not dry out in the incubator, before closing the petri dish. Place upright in incubator for the desired amount of time. The experiment can now proceed untouched for up to 24 hours (see Supplementary video).
Figure 2. Device with bacteria droplets, 1 droplet per habitat.
Figure 3. An ‘upright’ petri dish + kimwipes + device + coverslip ready to be incubated.
- Recovering from the devices (perform in a BSC): Open petri dish, carefully remove and discard kimwipes and then use sterile tweezers to gently unseal the device and place face up in the lid of the petri dish. By around 12 hours, the spaces between the habitats will be void of liquid from PDMS absorption, preventing microbes from being mixed across chambers during disassembly. Habitats that have dried out will appear white (Fig. 4) and cannot be used. Dip sterile inoculating loop into an eppendorf with PBS, then use that loop to scrape one of the habitats (Fig. 5). Dip that inoculating loop back into the eppendorf media again, which can then be grown overnight for further analyses such as plating for relative cell count (if there are different strains) and other molecular analyses. Repeat for the rest of the habitats that you are interested in, using new inoculating loops. The PDMS device can now be cleaned as in Step 1 and reused again.
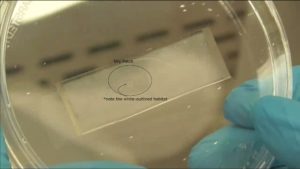
Figure 4. View of an inoculated and incubated device, looking through the bottom of a petri dish.
Figure 5. Recovering bacteria from a habitat in the disassembled PDMS device.
What else should I know?
The recovery technique can be used to estimate relative proportions of different types of microbes (e.g. morph frequencies) which is useful when performing competition assays and evolutionary experiments. Unlike in Tekwa et al. (2015), this technique forgoes the use of a confocal microscope; assessment of the contents of the device is instead performed through direct microbe recovery and standard plating procedures.
Links to Videos
These videos go through the specific procedure that we used to perform experiments on competition and cooperation in Pseudomonas aeruginosa and may be useful in determining specific amounts of media, growth times, etc. that may be utilized for experiments with similar PDMS microfluidic devices.
Part 1 – Intro + Washing the Devices https://youtu.be/Bne3ZN3wU4Q
Part 2 – Inoculating the Devices https://youtu.be/p-UYIRreYyM
Part 3 – Recovering from the Devices https://youtu.be/YLnmGxqMYgE
Supplementary – Fluorescent Bacteria Experiment https://youtu.be/lgMtryS62pA
Acknowledgements
AGr was supported by an NSERC Undergraduate Student Research Award and by an NSERC Discovery Grant. EWT was supported by the Fonds Québécois de la Recherche sur la Nature et les Technologies and the Québec Centre for Biodiversity Science. AGo was supported by the Canada Research Chair program and NSERC Discovery grants. DN was supported by a CFI Leaders Opportunity Fund (25636), a Burroughs Wellcome Fund CAMS award (1006827.01) and a CIHR salary award.
References
Cho, H., Jönsson, H., Campbell, K., Melke, P., Williams, J. W., Jedynak, B., … & Levchenko, A. (2007). Self-organization in high-density bacterial colonies: efficient crowd control. PLoS biology, 5(11), e302.
Connell, J. L., Ritschdorff, E. T., Whiteley, M., & Shear, J. B. (2013). 3D printing of microscopic bacterial communities. Proceedings of the National Academy of Sciences, 110(46), 18380-18385.
Folkesson, A., Jelsbak, L., Yang, L., Johansen, H. K., Ciofu, O., Høiby, N., & Molin, S. (2012). Adaptation of Pseudomonas aeruginosa to the cystic fibrosis airway: an evolutionary perspective. Nature reviews. Microbiology, 10(12), 841.
Hol, F. J., & Dekker, C. (2014). Zooming in to see the bigger picture: Microfluidic and nanofabrication tools to study bacteria. Science, 346(6208), 1251821.
Keymer, J. E., Galajda, P., Lambert, G., Liao, D., & Austin, R. H. (2008). Computation of mutual fitness by competing bacteria. Proceedings of the National Academy of Sciences, 105(51), 20269-20273.
Or, D., Smets, B. F., Wraith, J. M., Dechesne, A., Friedman, S. P. (2007). Physical constraints affecting bacterial habitats and activity in unsaturated porous media – a review. Advances in Water Resources, 30(6), 1505-1527.
Park, S., Wolanin, P. M., Yuzbashyan, E. A., Silberzan, P., Stock, J. B., & Austin, R. H. (2003). Motion to form a quorum. Science, 301(5630), 188-188.
Tekwa, E. W., Nguyen, D., Juncker, D., Loreau, M., & Gonzalez, A. (2015). Patchiness in a microhabitat chip affects evolutionary dynamics of bacterial cooperation. Lab on a Chip, 15(18), 3723-3729.
Tekwa, E.W., Nguyen, D., Loreau, M., Gonzalez, A. Defector clustering is linked to cooperation in a pathogenic bacterium. In review.