
We hear from Hui Luo, Georg Kastlunger, Ifan E. L. Stephens, and Magda Titirici about their groundbreaking study in the latest issue of EES Catalysis, titled ‘Selective glycerol to lactic acid conversion via a tandem effect between platinum and metal oxides with abundant acid groups‘ to gain deeper insight into their groundbreaking work.
What inspired you to focus on the selective conversion of glycerol to lactic acid?
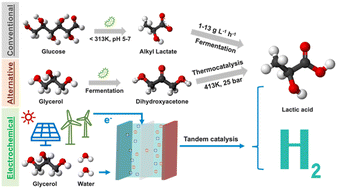
Hui: “The reaction of glycerol transformation is a very complex process. Dozens of different products can be formed, depending on the reaction conditions. It is important to identify the products that are of high-value and useful. Therefore, before starting the project we reviewed the glycerol product’s market in addition to receiving industrial feedback. This is how we learned that among all products, lactic acid has good value combined with relatively large market size and therefore we started to focus research on the selectively of converting glycerol to lactic acid.”
Can you explain the significance of using platinum and metal oxides in your catalyst design?
Hui: “Traditionally, Pt on carbon is used as the electrocatalyst for glycerol oxidation, while metal oxides are used in thermal catalysis. During our research we concluded that glycerol-to-lactic acid conversion is a combination of electrochemical + thermochemical catalysis. Hence, we took inspirations from both research fields and designed a tandem catalytic system, which doubled the lactic acid selectivity. Such discovery shows the importance of thinking “out of the box” and seeking inspiration from different research fields.”
What were the biggest challenges you faced during this research?
Hui: “The biggest challenge we faced was identifying the intermediate compound, dihydroxyacetone. Because it is highly unstable in an alkaline environment, we were not able to probe it directly. However, combining our experiments with theoretical calculations, we were able to prove the tandem effect happening at the Pt and metal oxide interface. We are actively working on resolving this challenge by combining different in-operando techniques, which will be shared in our future works.”
How did you determine the optimal conditions for the tandem effect between platinum and metal oxides?
Georg: “We already knew that platinum under alkaline conditions is an excellent catalyst for glycerol electrooxidation. To achieve improved selectivity towards lactic acid, our initial hypothesis, based on literature for thermal catalysis, was that we would need acidic catalytic sites.
We probed our hypothesis by investigating composite catalysts based on a combination of metal oxides with different acidic strengths and Pt. We performed theoretical calculations based on quantum mechanical simulations to investigate the catalytic conversion step by step. This way we could prove the correlation of selectivity towards lactic acid with the acidity by defining simplified descriptors for screening for optimised catalysts.”
Could you elaborate on the key findings of your study?
Georg and Hui: “Platinum is the most investigated catalyst for the oxidation of glycerol. However, its metallic nature favours the pure electrochemical oxidation path to highly oxidized products such as glyceric acid and other small organic acids. Lactic acid is a partially oxidized C3 product which necessitates a series of intramolecular conversions when produced from glycerol. Thus, to achieve improved selectivity towards lactic acid, we need to “isolate” partially oxidized intermediates from the Pt active sites and provide catalytic sites for the non-electrochemical conversion, such as oxides rich in Lewis acid groups. To tune the ability of capturing intermediate products, the oxide’s binding affinity can be optimized by increasing the acidity of its sites. This tuning factor, however, with the competing tendency for oxidation of the oxide’s active sites, correlating with their acidity as well.
Combining experimental and theoretical results, we have elucidated the reaction mechanism demonstrating that Pt alone has a poor selectivity towards lactic acid due to its tendency to undergo a pure electrochemical process that produce a mixture of acids. Furthermore, we demonstrated that the surface acidity of certain metal oxide catalysts is crucial for shifting the reaction towards the dehydration pathway occurring via binding and catalysing the dihydroxyacetone intermediate.”
How does your work improve the efficiency and selectivity of lactic acid production compared to previous methods?
Hui: “By benchmarking our performance with previous methods, we can say that our selectivity is among the highest reported so far. Recently there are a few more studies in this field that combine precious metal catalysts and metal oxides to improve lactic acid selectivity, and we are glad to see our hypothesis validated and proven by other researchers.”
What potential applications do you see for your findings in the industry?
Ifan: “Lactic used in its lactide form is the precursor for producing poly(lactic) acid which is the most used biodegradable plastic for consumer goods. Hui conducted a high level technoeconomic analysis, which showed the superiority of electrochemical methods vs fermentation for lactic acid production, provided that renewable energy is used. In addition, the coproduction of hydrogen on the positive electrode increases the economic value of the overall process. Apart from evolving hydrogen, the glycerol electrooxidation could also be coupled with other electroreduction reactions such as CO2 to ethylene, yielding increased economic value.”
Are there any future research directions or improvements you are planning to explore?
Ifan and Magda: “We plan to improve the selectivity towards lactic acid at higher current densities further by using different substrates such as nickel foam. In addition, more direct spectroscopic proof of the reaction pathway that leads to lactic acid production on acidic sites would allow us to develop even more targeted synthesis routes. It would also be useful to get a better understanding and control of mesocopic effects relative to surface catalysis.”
What impact do you hope your research will have on the field?
Magda: “The impact will be in the academic community working on waste to chemicals and in electrocatalysis, on alcohols electrooxidation. It has generated new knowledge on factors controlling the selectivity of alcohol oxidations to partially oxidised products and the knowledge could be translated to other, more complex biomass oxidation reactions of interest for a circular economy and biobased products.”

 |
How do we discover new energy solutions?
EES Catalysis publishes high quality research on energy and environmental catalysis. It delivers the same impact and influence that researchers associate with our Energy & Environmental Science brand. As a multidisciplinary platform, the journal covers catalysis in all subject areas like chemistry, materials science and engineering. At its core, EES Catalysis aligns with the UN’s Sustainable Development Goal (SDG) 7 – to ensure access to affordable, reliable and modern energy for all.
Find out more https://rsc.li/EESCatalysis |

