Tom Bond is a Junior Research Fellow (JRF) in the Environmental and Water Resource Engineering (EWRE) section at Imperial College London. His research is focused on the role aquatic chemistry can play in identifying and removing hazardous contaminants during water and wastewater engineering treatment processes.
Tom Bond is a Junior Research Fellow (JRF) in the Environmental and Water Resource Engineering (EWRE) section at Imperial College London. His research is focused on the role aquatic chemistry can play in identifying and removing hazardous contaminants during water and wastewater engineering treatment processes.
Although he has spent most of his research career in engineering departments, his first degree was in chemistry and he is interested in synergistic interactions between the two disciplines. He holds a PhD on the treatment of disinfection byproduct precursors and MSc in Water and Wastewater Engineering, both from Cranfield University, and a first class honours degree (MSci) in chemistry from Bristol University.
Read Tom’s Emerging Investigators article ‘formation of disinfection byproducts during the preparation of tea and coffee’ here.
– How has your research evolved from your first to your most recent article?
My first article was on disinfection byproducts, as is this latest one. However, in between I have also worked on lots of different areas, so hopefully I am now knowledgeable about a wider range of research topics then when I started in research.
– What aspect of your research are you most excited about at the moment?
In general, I am excited by having the freedom to work on different topics and pursue things which interest me. If I am able to make any contributions to improving the public health impact of drinking water then that would be even better.
– Your paper discusses an issue that potentially affects most of us. How did you come up with this idea?
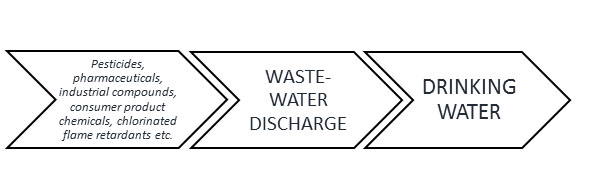
I was reading a thesis from a student at Imperial College, in which was made an incidental comment that tea and coffee represent potential sources of disinfection byproducts. And I thought, That’s an interesting idea actually. After looking in the literature, I was quite surprised to find that this was not something that had been looked at much detail previously. This made it seem like something that would be worth proposing as an MSci research project, which turned out to be the case, especially as an excellent student selected it (Seeheen Tang).
– What was your biggest challenge during this research?
We had some technical challenges with the laboratory work along the way. The biggest was that I initially wanted to undertake a liquid-liquid extraction on the chlorinated tea and coffee samples prior to analysing for disinfection byproducts by gas chromatography. This is a standard method in the drinking water research.
However, as we found out, when you try extracting tea or coffee into an organic solvent you get a horrible frothy mess, which is hopeless for extracting anything from. After trying some alternatives, I ended up sending some samples to an external lab for trihalomethane analysis using a headspace gas chromatography method. This relies on heating the sample to separate the volatile trihalomethanes, rather than extracting them.
– How did you find out about Environmental Science: Water Research & Technology and how was your experience?
It was mentioned to me by colleagues at Imperial, while I have also seen it advertised at conferences I have attended. The editorial and peer review process was very efficient in the case of this article.
– How do you spend your spare time?
In no particular order: walking/hiking, going to the pub, eating out and birdwatching. The last of these is my biggest passion, even if I probably spend more time on the other activities mentioned.
– If you could not be a scientist, but could be anything else, what would you be?
A writer, although I am not sure of which type. Of the various enjoyable aspects of working in a university, writing is the part I like most. And it would intrigue me to try writing in a different style to that required by science/academia. As a secondary alternative, being a professional footballer would be ok (!), although I fear that I am already too old, not to mention unskilled, for that possibility.
– Can you share one piece of career-related advice or wisdom with other early career scientists?
Try to improve a variety of aspects of your CV, rather than concentrating on one or two, as this should give you more opportunities in the future. It also helps if you know where you want to go in your career, as then you can plan strategically what is needed to get there.















![By Matthew Bowden www.digitallyrefreshing.com (http://www.sxc.hu/photo/158242) [Attribution], via Wikimedia Commons Running water from tap](https://upload.wikimedia.org/wikipedia/commons/7/73/Wasserhahn.jpg)