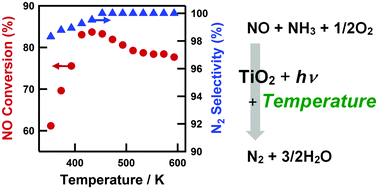
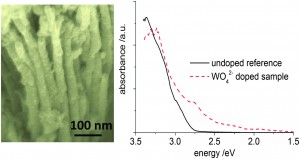
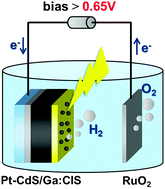
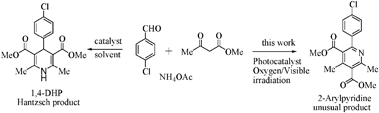
Alkene hydration (the addition of water to a carbon double bond) is an important industrial process for the direct synthesis of alcohols, widely used as chemical intermediates for resins, agricultural chemicals, surfactants, etc. In an extension of their previous work which involved selective aromatic ring hydroxylation by water with a platinum-loaded titanium oxide photocatalyst, the Yoshida Group, applied this photocatalytic system to alkene hydration.
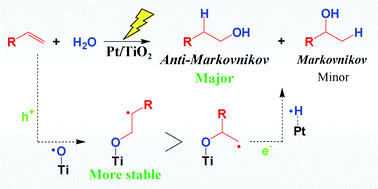
The Pt–TiO2 photocatalyst efficiently promoted anti-Markovnikov hydration of various alkenes with high selectivity. A possible reaction mechanism is also discussed in detail based on experiment and molecular orbital calculations. The results should prove useful for designing novel photocatalysts for selective organic transformations.
Read more about these developments in photocatalytic research by downloading the full article now:
Anti-Markovnikov hydration of alkenes over platinum-loaded titanium oxide photocatalyst
Hayato Yuzawa, Shoko Yoneyama, Akihito Yamamoto, Masanori Aoki, Kazuko Otake, Hideaki Itoh and Hisao Yoshida
Catal. Sci. Technol., 2013, DOI: 10.1039/C3CY00019B
This article is part of a themed issue on photocatalysis that is due to be published later this year.
Also, check out the previous paper which sparked this research:
Photocatalytic hydroxylation of aromatic ring by using water as an oxidant
Hisao Yoshida, Hayato Yuzawa, Masanori Aoki, Kazuko Otake, Hideaki Itoh and Tadashi Hattori
Chem. Commun., 2008, 4634-4636
DOI: 10.1039/B811555A
And the latest work by this group:
Heterogeneous palladium catalyst hybridised with titanium oxide photocatalyst for direct C–C bond formation between aromatic ring and acetonitrile
Hisao Yoshida, Yuki Fujimura, Hayato Yuzawa, Jun Kumagai and Tomoko Yoshida
Chem. Commun., 2013, Accepted Manuscript
DOI: 10.1039/C3CC41068D