 In the field of heterogeneous catalysis, the characteristics of the support can make a substantial difference in the operating conditions of a system and on its performance. Boron nitride (BN), in its hexagonal, graphite-like morphology, has proven to be a promising support for a number of catalysts and processes, including the synthesis of ammonia over barium/ruthenium catalysts thanks also to its higher chemical and thermal stability.
In the field of heterogeneous catalysis, the characteristics of the support can make a substantial difference in the operating conditions of a system and on its performance. Boron nitride (BN), in its hexagonal, graphite-like morphology, has proven to be a promising support for a number of catalysts and processes, including the synthesis of ammonia over barium/ruthenium catalysts thanks also to its higher chemical and thermal stability.
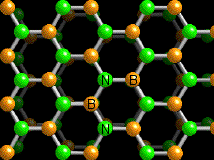
Chinese researchers led by Liqiang Xu focused on the synthesis of such supports, developing ultrathin nanosheets (<20 layers) with high specific surface area that could potentially be complementary to their carbon counterparts.
The group developed a convenient solid phase synthesis starting from boron oxide, zinc powder and N2H4 2HCl that yielded nanosheets with thickness varying from 2 to 6 nm. Notably, when zinc was absent, no BN was produced. Replacing zinc with iron or manganese resulted in lower yields or thicker nanosheets.
To test the properties of the new boron nitride support, the nanosheets were impregnated with platinum and employed in the oxidation of carbon monoxide with interesting results. Not only did the system reached almost full conversion with catalyst concentration as low as 2, 1 and 0.04%, but the respective temperature at which it was achieved (respectively 165, 210 and 310 °C) was lower than with conventional BN or alumina supports.
The material was also functionalised with gold nanoparticles, but to obtain interactions strong enough to anchor them to the surface, a supplementary functionalisation of the material was required. After hydrothermal treatment of the nanosheet with hydrogen peroxide, the Au nanoparticles dispersed more efficiently and allowed for higher loading.
In the Authors` view, these Au/BN nanosheet could find applications in optical materials, sensors and photocatalysis.
Find the full communication here.
Convenient synthesis and applications of gram scale boron nitride nanosheets
Liancheng Wang, Changhui Sun, Liqiang Xu and Yitai Qian
Catal. Sci. Technol., 2011, Advance Article
DOI: 10.1039/C1CY00191D