High nucleophilic reactivity and functional group tolerance are opposing properties of organometallic compounds. While organolithium compounds are highly nucleophilic so react with most electrophiles, organosilicon compounds are on the low reactivity end of the nucleophilicity scale. They tolerate numerous electrophilic functional groups and often require (Lewis) base activation to react.
Organoboron compounds, which are key intermediates in organic synthesis, are positioned in between. While some of them undergo non-catalysed C–C bond forming reactions with various electrophiles, others tolerate a variety of functional groups and are most valuable substrates for Pd-catalysed cross couplings (Suzuki–Miyaura reactions).
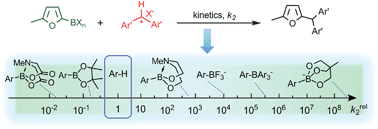
As it is difficult for the preparative chemist to decide which functional groups are tolerated and which do react with organoboron reagents in the absence of a catalyst, scientists in Germany have established a quantitative reactivity scale for representatives of the most important classes of organoboron reagents and they have applied these data for predicting the feasibility of transition metal free C–C bond forming reactions.
Reference:
Nucleophilicity parameters for designing transition metal-free C-C bond forming reactions of organoboron compounds
G Berionni, B Maji, P Knochel and H Mayr, Chem. Sci., 2012
DOI: 10.1039/c2sc00883a