On 19th – 22nd January 2024, the Bioinorganic Chemistry Gordon Research Seminar (GRS) took place. This annual seminar provides an opportunity for young doctoral and post-doctoral researchers to present and discuss their work. GRS encourages active participation and engaged discussion from all attendees, in order to encourage scientific collaboration.
This year, the conference focused on Elucidating the Influence of Metals on Biological Processes and RSC Advances were lucky enough to sponsor the event and support attendance of early career researcher! Caitlin Palmer is a graduate student at Northwestern University, United States. She completed her BS in Chemistry and Biochemistry and MS in Inorganic Chemistry at East Carolina University.
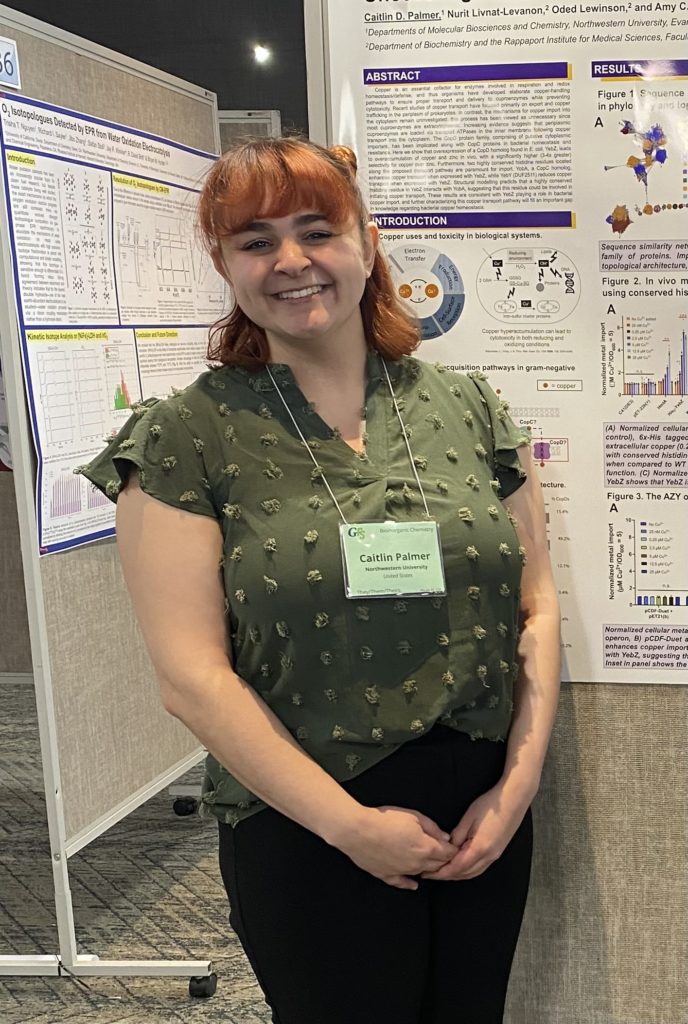
Caitlin Palmer
At the conference, Caitlin presented her research on “Uncovering the Role of CopD in Methanotroph Copper Homeostasis”.
Caitlin has told us about her research and discussed the triumphs and challenges she has faced throughout her career so far. She also provided some advice for other students and early career researchers.
What is the focus of your research and why it is of current interest?
The focus of my research is how metals are trafficked, stored, and delivered to essential enzymes in bacteria. Specifically, I work on a new class of copper transport proteins that are only found in bacteria, and potentially deliver copper to enzymes involved in carbon metabolism and antibiotic resistance pathways. Because of this, my lab is very interested in studying the mechanism of how this class of proteins is involved in copper homeostasis, and how they can be targeted for drug therapies down the line.
What are the key design considerations for your study?
The key design considerations for my study include finding ample controls for some of the in vivo assays I’ve created to monitor a copper transport in bacteria. In addition, since this class of proteins is a membrane protein, it’s been very important to optimize its stable expression and purification for in vitro assays.
Which part of the research so far has proved to be the most challenging?
I think the most challenging portion of my research so far was the switch to membrane proteins – during my undergraduate and masters, I worked with soluble metal binding proteins that had been established in the literature for many years, which were relatively easy to purify and characterize for my experiments. Now in my PhD, working with membrane proteins has been a challenge due to their solubility and instability issues during purification. I’ve had to work with numerous constructs (+30 homologs and tag configurations) in order to optimize stable expression and purification of these proteins.
What aspect of the work are you most excited about?
I think I’m most excited about the structural aspect of my project – I’ve been working on collecting cryoEM data recently, so I am very excited to start my foray into structural characterization, especially since cryoEM is such a hot field right now.
What advice would you give to students and early career researchers in a similar situation to yourself?
The best advice I was given came from a postdoctoral scholar in our lab – she told me that the best way to get through some of the lows and “failed experiments” in grad school is to keep your energy and excitement up throughout it, even when it feels like everything is going horribly. My project has been really difficult, and at times when I’ve felt like nothing is going right, I’ve really leaned into this thought of staying excited about my research and why it is so exciting for me to do.
What are some of the challenges you have faced as a researcher starting out in your career and what positive progress have you seen throughout your career so far?
Since I’m a graduate student, I’m not too far into my career just yet, but at least from my transition from undergraduate to graduate studies, I’ve noticed my confidence in my research and my technical abilities has really increased – during my undergraduate research experience, I had the unfortunate experience of being viewed as only a tech and someone who only ran data for others (rather than contributing to science and doing thoughtful experiments), so that really hurt my confidence in my abilities. However, as I’ve grown my skills and spent more time in the lab as a graduate student, I’ve gained more confidence and respect as a researcher, which has been a very restorative experience.
Do you have any recommendations for improving the STEM workforce to create an environment that better supports early career researchers? Is there anything publishers such as the RSC can do to help?
I think the biggest advice I can give is to provide more funding and support for early career researchers, especially from minority and LGBT/queer communities, because these researchers represent more of the type of graduate students and postdoctoral scholars that are being attracted to STEM careers, including myself. One of the biggest influences on my career was an early career faculty member at my undergraduate institution who is queer and a minority – seeing him thrive in the face of adversity really shaped my perspective as a young queer researcher, and gave me the spark to love my research even more and continue on my graduate school career. To the second part of your question, I think journals and publishers should strive to publish articles and thought pieces from a diverse range of researchers, and really try to support these historically underrepresented communities (in the form of publishing, scholarships, travel awards, funding, etc) to help them achieve greater success. It really takes a village to support early career researchers, so any support from the community is paramount to their success.
Anything else to note?
I also love to plug my cat in any interview I do, so please find attached to this email a copy of my cat (Miss Sammie) who keeps me sane during graduate school. She has also donated many whiskers to our lab for crystallography experiments, so she is an active “researcher” in my lab as well!

Miss Sammie

Submit to RSC Advances today! Check out our author guidelines for information on our article types or find out more about the advantages of publishing in a Royal Society of Chemistry journal.
Keep up to date with our latest Popular Advances, Reviews, Collections & more by following us on Twitter. You can also keep informed by signing up to our E-Alerts.
Comments Off on Bioinorganic Chemistry (GRS) – Interview with Caitlin Palmer