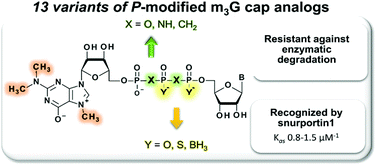
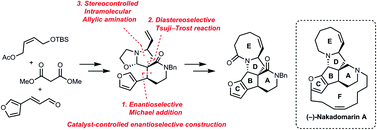
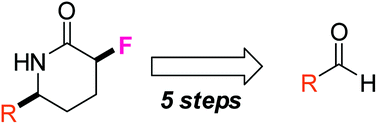
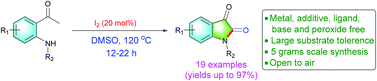
Organic and Biomolecular Chemistry (OBC) is delighted to announce a high-profile web themed issue on Contemporary Synthetic Chemistry in Drug Discovery. The guest editors for this issue are Professor Angela Russell (University of Oxford, UK), Professor Douglas E. Frantz (The University of Texas at San Antonio, USA), Dr Matthew Duncton (Rigel, San Francisco, USA), Dr Graham Wynne (University of Oxford, UK) and Dr Shane Krska (Merck, Boston, USA).
Deadline for Submission: 30 April 2016
If you are interested in taking part in this issue, please email OBC: obc-rsc@rsc.org
Manuscripts can be submitted using the Royal Society of Chemistry’s online article submission service. Please clearly state that the manuscript is submitted for the themed issue on Contemporary Synthetic Chemistry in Drug Discovery.
The level of quality of this issue will be high, and all manuscripts will undergo the journal’s normal peer review process. Guidelines are available at rsc.li/1K0EgYx and rsc.li/1OoQWQh
Scope of the issue
This issue aims to demonstrate the importance of synthetic chemistry in drug discovery. Specifically it will highlight how synthetic methods can be used to address hurdles currently being faced in modern medicinal chemistry. Areas falling within the scope of the issue include:
• CH-Functionalization, including late-stage CH-functionalization & CH-functionalization via biomimetic catalysis
• Fluorination chemistry, including late-stage fluorination, CF3, SCF3 & SF5 chemistry
• Asymmetric chemistry, including use of organocatalytic methods
• Synthesis of compounds with enhanced 3-dimensional character (including cross-coupling & other metal-catalyzed reactions, particularly in relation to sp3-functionalization)
• Modern heterocyclic chemistry including synthesis of novel heterocycles, highly-decorated heterocycles & synthesis/use of small heterocyclic groups (e.g. oxetanes, azetidines, etc.)
• Modern synthetic techniques, including flow chemistry, automated synthesis, high-throughput synthesis, microwave-assisted chemistry, multi-component reactions & sustainable chemistry
• Synthesis/modification of natural products, or natural-product-like compounds (diversity-orientated synthesis)
• Synthesis utilizing enzymatic chemistry and its use in drug discovery
• The use of novel reagents/reactions in drug discovery
All submissions should clearly show/explain what challenge within drug discovery is being addressed by the presented work. Articles covering general organic synthesis without this connection are not suitable for this issue.
Inclusion of biological data is not a requirement for submission but you are welcome to include any such data that you feel supports your article. Please note that the main focus of the manuscript should be on the organic synthesis and not the medicinal chemistry.
To view recent articles or find out more about OBC, please visit the journal’s homepage:
Organic & Biomolecular Chemistry – Rapid publication of high quality organic chemistry research












 Organic synthesis is often heralded as more art than science. An organic chemist’s eye for complexity, breaking down structures into simpler forms, is honed and nurtured over decades. But, is it possible to take this seemingly intangible skill and quantify it, putting a simple number on how complex a chemical structure actually is?
Organic synthesis is often heralded as more art than science. An organic chemist’s eye for complexity, breaking down structures into simpler forms, is honed and nurtured over decades. But, is it possible to take this seemingly intangible skill and quantify it, putting a simple number on how complex a chemical structure actually is?



![Stereoselective intermolecular [2 + 2]-photocycloaddition reactions of maleic anhydride: stereocontrolled and regiocontrolled access to 1,2,3-trifunctionalized cyclobutanes Stereoselective intermolecular [2 + 2]-photocycloaddition reactions of maleic anhydride](http://pubs.rsc.org/services/images/RSCpubs.ePlatform.Service.FreeContent.ImageService.svc/ImageService/image/GA?id=C4OB01383B)