This article is part of a forthcoming themed issue on self assembly in inorganic chemistry, with Guest Editors Paul Kruger (University of Canterbury) and Thorri Gunnlaugsson (Trinity College Dublin).
Read more for FREE about these developments in self assembly at:
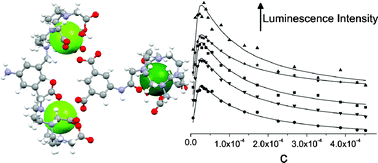
Self-assembly between dicarboxylate ions and a binuclear europium complex: formation of stable adducts and heterometallic lanthanide complexes
James A. Tilney, Thomas Just Sørensen, Benjamin P. Burton-Pye and Stephen Faulkner
Dalton Trans., 2011, Advance Article
DOI: 10.1039/C1DT11103E










