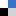We are very pleased to announce that Professor Lichen Yin has been appointed as an Associate Editor for Biomaterials Science.
 |
| Lichen Yin is a Professor in the Institute of Functional Nano & Soft Materials (FUNSOM), Soochow University. His research interest is focused on the engineering of polymeric and/or bio-inspired nanomaterials that can overcome multiple physiological barriers and deliver biomacromolecular drugs such as nucleic acids, proteins, and peptides. His research works to create new technologies and therapeutics for the treatment of cancer, cardiovascular diseases, autoimmune diseases, etc. He is the recipient of the National Science Fund for Distinguished Young Scholars of China (NSFC, 2023), and is the “Distinguished Professor’’ of Jiangsu Province. |
Submit your manuscript to Professor Yin
Read Professor Yin’s Biomaterials Science articles:
| One-pot synthesis of dynamically cross-linked polymers for serum-resistant nucleic acid delivery
Zhisong Sun, Mengyao Ren, Bingchen Shan, Qiang Yang, Ziyin Zhao, Xun Liu and Lichen Yin Biomater. Sci., 2023, 11, 5653-5662 |
 |
 |
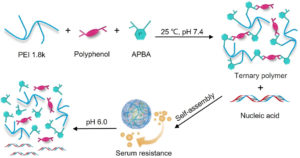
Spherical α-helical polypeptide-mediated E2F1 silencing against myocardial ischemia-reperfusion injury (MIRI)
Rongying Zhu, Mengying Hou, Yang Zhou, Huan Ye, Lei Chen, Chenglong Ge, Shanzhou Duan, Lichen Yin and Yongbing Chen |
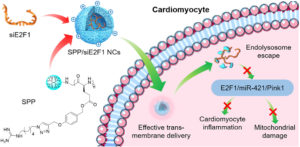
| A near-infrared light-controlled, oxygen-independent radical generating nano-system toward cancer therapy
Desheng Cao, Hua He, Wei Li, Jin Yan, Jianhua Wu, Mengyuan Yin, Yang Zhou, Zhuchao Zhou and Lichen Yin Biomater. Sci., 2021, 9, 4054-4065 |
 |
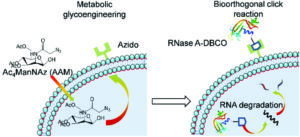 |
Cytosolic protein delivery via metabolic glycoengineering and bioorthogonal click reactions
Ziyin Zhao, Zhimin Zhang, Shanzhou Duan, Xun Liu, Renxiang Zhou, Mengying Hou, Yonghua Sang, Rongying Zhu and Lichen Yin Biomater. Sci., 2021, 9, 4639-4647
|
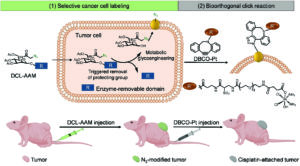
| Cancer cell-targeted cisplatin prodrug delivery in vivo via metabolic labeling and bioorthogonal click reaction
Xun Liu, Fan Wu, Kaimin Cai, Ziyin Zhao, Zhimin Zhang, Yongbing Chen, Yong Liu, Jianjun Cheng and Lichen Yin Biomater. Sci., 2021, 9, 1301-1312 |
 |
All the highlighted articles are currently FREE to read until January 31st 2024!